Is Covalent Bond Between Two Nonmetals
Muz Play
Apr 01, 2025 · 6 min read
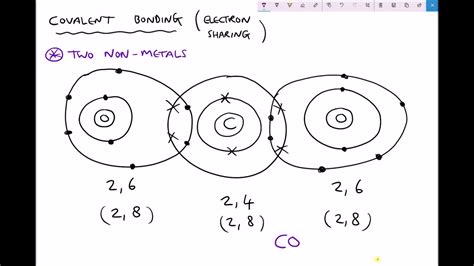
Table of Contents
Is a Covalent Bond Between Two Nonmetals? A Deep Dive into Chemical Bonding
The simple answer is yes, a covalent bond is almost always formed between two nonmetal atoms. Understanding why requires delving into the fundamental principles of atomic structure and chemical bonding. This comprehensive guide will explore the nature of covalent bonds, the characteristics of nonmetals, and the reasons behind the strong preference for covalent bonding between these elements. We'll also touch upon exceptions and nuanced situations that might challenge this general rule.
Understanding Covalent Bonds: Sharing is Caring
A covalent bond is a chemical bond formed by the sharing of electron pairs between atoms. This sharing allows each atom to achieve a more stable electron configuration, often resembling the electron configuration of a noble gas (a highly stable group of elements). Unlike ionic bonds, which involve the transfer of electrons, covalent bonds result in a more equal distribution of electrons between the bonded atoms.
The Role of Valence Electrons
The key players in covalent bond formation are valence electrons. These are the electrons located in the outermost shell of an atom. Atoms strive to achieve a full outermost shell, often referred to as an octet (eight electrons), which provides exceptional stability. By sharing valence electrons, atoms effectively "borrow" electrons from each other to complete their outermost shells.
Examples of Covalent Bonds
Simple examples illustrate this process:
- Hydrogen (H₂): Two hydrogen atoms, each with one valence electron, share their electrons to form a single covalent bond (H-H). Each hydrogen atom effectively achieves a full shell of two electrons (like helium).
- Oxygen (O₂): Two oxygen atoms, each with six valence electrons, share two pairs of electrons to form a double covalent bond (O=O). Each oxygen atom now has eight electrons in its outermost shell.
- Water (H₂O): Oxygen shares electron pairs with two hydrogen atoms, forming two single covalent bonds. Oxygen achieves an octet, and each hydrogen atom achieves a duet (two electrons).
The Nature of Nonmetals
Nonmetals are elements that generally lack metallic properties. They are typically poor conductors of heat and electricity, are brittle in their solid state, and often exist as gases or low-melting-point solids at room temperature. Their chemical behavior is largely characterized by their tendency to gain electrons to achieve a stable electron configuration.
Electronegativity and Nonmetals
Nonmetals generally have high electronegativity. Electronegativity is a measure of an atom's ability to attract electrons towards itself in a chemical bond. Because nonmetals are closer to achieving a stable noble gas configuration by gaining electrons, they strongly attract electrons involved in covalent bonding. This strong attraction leads to the sharing of electrons rather than the outright transfer seen in ionic bonds.
Why Covalent Bonds Favor Nonmetals
The combination of high electronegativity and the need to gain electrons to achieve stability makes covalent bonding a highly favorable interaction between nonmetal atoms. Here's a breakdown:
- Similar Electronegativities: Nonmetals tend to have similar electronegativities. If the electronegativity difference between two atoms is large (as is the case between a metal and a nonmetal), electrons are transferred, resulting in an ionic bond. However, the relatively small electronegativity differences between nonmetals promote electron sharing instead of transfer.
- Electron Affinity: Nonmetals have a high electron affinity, meaning they readily accept electrons to fill their outer electron shells. The sharing of electrons in a covalent bond is a more efficient way for nonmetals to satisfy this affinity than complete electron transfer.
- Octet Rule: The octet rule is a guideline that dictates that atoms tend to form bonds to achieve a full outer shell of eight electrons. Covalent bonding allows nonmetals to achieve this stable octet configuration by sharing electrons with each other.
Exceptions and Nuances
While the rule that covalent bonds occur between nonmetals is a very good guideline, there are some exceptions and nuances to consider:
Polar Covalent Bonds
Not all covalent bonds are equal. In polar covalent bonds, the shared electrons are not equally shared. This happens when the electronegativity difference between the two nonmetals is significant, but not large enough to cause a complete transfer of electrons. One atom will have a slightly more negative charge (δ-), and the other atom will have a slightly more positive charge (δ+). Water (H₂O) is a classic example of a molecule with polar covalent bonds.
Coordinate Covalent Bonds
Also known as dative bonds, these covalent bonds involve one atom providing both electrons for the shared pair. This is common in complex ions and molecules containing transition metals. While it's still a covalent bond, the electron pair originates from a single atom.
Metallic Character and Nonmetals
Some nonmetals, particularly those near the metalloid boundary on the periodic table (like silicon and selenium), can exhibit some degree of metallic character. This might lead to bonding that shows aspects of both covalent and metallic bonding.
Hydrogen Bonding
Hydrogen bonding is a special type of intermolecular force (not a true chemical bond), but it’s crucial in many biological and chemical systems. It arises from the strong attraction between a hydrogen atom bonded to a highly electronegative atom (like oxygen or nitrogen) and another highly electronegative atom in a nearby molecule. While not a covalent bond between two nonmetals, it plays a significant role in the properties of many substances.
Advanced Concepts and Applications
Understanding the nuances of covalent bonding is crucial in various scientific fields:
Organic Chemistry
Covalent bonding is the cornerstone of organic chemistry, the study of carbon-containing compounds. The ability of carbon to form strong covalent bonds with itself and other nonmetals (like hydrogen, oxygen, and nitrogen) leads to the vast diversity of organic molecules found in living organisms and synthetic materials.
Biochemistry
Covalent bonds form the backbone of biological macromolecules like proteins, carbohydrates, and nucleic acids. The specific types of covalent bonds and their arrangements determine the structure and function of these molecules, critical for life processes.
Materials Science
Covalent bonding is essential in understanding the properties of many materials, from semiconductors (like silicon) to polymers (like plastics). The strength and nature of the covalent bonds influence the hardness, conductivity, and other properties of these materials.
Molecular Modeling and Simulation
Computational methods are used extensively to study covalent bonds and predict the properties of molecules. These simulations are essential for drug discovery, materials design, and understanding chemical reactions.
Conclusion
In summary, covalent bonds are primarily formed between two nonmetal atoms. This preference arises from the similar electronegativities, the desire to gain electrons to achieve noble gas configuration, and the efficiency of electron sharing in achieving stable octets. While exceptions and nuanced bonding situations exist, understanding the fundamental principles of covalent bonding is crucial for comprehending the structure, properties, and behavior of a wide range of substances in various fields of science and technology. This deep understanding helps us unlock the secrets of the molecular world and harness the power of chemical bonding for advancements across numerous disciplines.
Latest Posts
Latest Posts
-
Sodium Acetate And Acetic Acid Buffer Equation
Apr 02, 2025
-
How To Add Radical Expressions With Variables
Apr 02, 2025
-
Give The Iupac Name Of The Carboxylic Acid Below
Apr 02, 2025
-
What Is An Abbreviated Electron Configuration
Apr 02, 2025
-
Difference Between A Strong Base And A Weak Base
Apr 02, 2025
Related Post
Thank you for visiting our website which covers about Is Covalent Bond Between Two Nonmetals . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
