Is The Hydrolysis Of Atp Endergonic Or Exergonic
Muz Play
Apr 02, 2025 · 5 min read
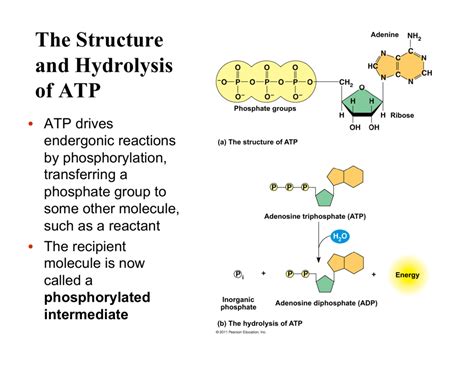
Table of Contents
Is the Hydrolysis of ATP Endergonic or Exergonic? A Deep Dive into Cellular Energetics
The hydrolysis of ATP, adenosine triphosphate, is a fundamental process in all living organisms. It fuels countless cellular reactions, powering everything from muscle contraction to protein synthesis. Understanding whether this reaction is endergonic (requiring energy) or exergonic (releasing energy) is crucial to grasping the basics of cellular metabolism and bioenergetics. The short answer is: the hydrolysis of ATP is exergonic. However, a complete understanding requires exploring the underlying thermodynamics, the role of ATP in coupled reactions, and the broader context of cellular energy transfer.
Understanding Endergonic and Exergonic Reactions
Before delving into the specifics of ATP hydrolysis, let's establish a clear understanding of endergonic and exergonic reactions. These terms describe the energy changes associated with chemical reactions:
-
Exergonic Reactions: These reactions release energy. The products have less free energy than the reactants. The change in Gibbs free energy (ΔG) is negative. Think of it like a ball rolling downhill – energy is released as it goes down. Many catabolic processes, such as the breakdown of glucose, are exergonic.
-
Endergonic Reactions: These reactions require energy input to proceed. The products have more free energy than the reactants. The change in Gibbs free energy (ΔG) is positive. This is like pushing a ball uphill – energy needs to be added to move it upwards. Anabolic processes, such as protein synthesis, are endergonic.
The Hydrolysis of ATP: An Exergonic Powerhouse
The hydrolysis of ATP involves the breaking of a phosphate bond in ATP to yield ADP (adenosine diphosphate) and inorganic phosphate (Pi). The reaction can be represented as:
ATP + H₂O ⇌ ADP + Pi
This reaction is exergonic, meaning it releases energy. The standard free energy change (ΔG°) for this reaction is approximately -30.5 kJ/mol under standard conditions. This negative ΔG° signifies that the reaction is spontaneous under standard conditions, meaning it favors the formation of products (ADP and Pi).
Why is ATP Hydrolysis Exergonic?
The exergonic nature of ATP hydrolysis stems from several factors:
-
Resonance Stabilization: ADP and Pi are more resonance-stabilized than ATP. Resonance structures represent different ways electrons can be distributed within a molecule. The increased stability of the products contributes to the lower energy state compared to the reactant, ATP.
-
Electrostatic Repulsion: The three negatively charged phosphate groups in ATP are closely packed together, resulting in significant electrostatic repulsion. Hydrolysis relieves this repulsion, contributing to the release of energy.
-
Hydration: ADP and Pi are better hydrated than ATP. The increased hydration of the products further stabilizes them and contributes to the overall negative ΔG.
ATP as the Cellular Energy Currency
ATP isn't just an exergonic molecule; it's the central energy currency of the cell. Its hydrolysis doesn't directly power most cellular processes; instead, the released energy is coupled to endergonic reactions. This coupling is crucial for life.
Coupled Reactions: Linking Exergonic and Endergonic Processes
Coupled reactions are essential for cellular function. An exergonic reaction, like ATP hydrolysis, can drive an endergonic reaction if they are coupled through a shared intermediate. The energy released from the exergonic reaction is used to "power" the endergonic reaction, making it proceed.
A classic example is the synthesis of glutamine from glutamate and ammonia:
Glutamate + NH₃ → Glutamine (Endergonic, ΔG° > 0)
This reaction is coupled to the hydrolysis of ATP:
ATP + H₂O → ADP + Pi (Exergonic, ΔG° < 0)
The overall coupled reaction has a negative ΔG, making it spontaneous:
Glutamate + NH₃ + ATP + H₂O → Glutamine + ADP + Pi
Factors Affecting ATP Hydrolysis
While the hydrolysis of ATP is generally exergonic, the actual free energy change (ΔG) can vary depending on several factors:
-
Concentration of Reactants and Products: The actual ΔG is influenced by the concentrations of ATP, ADP, and Pi within the cell. If the ATP concentration is high and the ADP and Pi concentrations are low, the ΔG will be more negative, favoring ATP hydrolysis.
-
Temperature and pH: Temperature and pH also affect the ΔG of the reaction. Changes in these conditions can alter the stability of ATP and its hydrolysis products.
-
Enzymes: Enzymes significantly affect the rate of ATP hydrolysis, but they do not change the ΔG of the reaction. They provide a lower activation energy pathway for the reaction to occur.
Beyond ATP Hydrolysis: Other Phosphate Transfers
While ATP hydrolysis is the most common energy source in the cell, other phosphate transfer reactions play important roles:
-
Phosphocreatine: In muscle tissue, phosphocreatine acts as a high-energy phosphate reserve, transferring its phosphate group to ADP to regenerate ATP.
-
GTP (Guanosine Triphosphate): GTP is another high-energy molecule that participates in various cellular processes, often involved in signal transduction and protein synthesis. Its hydrolysis is also exergonic.
The Importance of Understanding ATP Hydrolysis
A thorough understanding of ATP hydrolysis is fundamental to understanding cellular metabolism and numerous biological processes. This knowledge underpins our comprehension of:
-
Cellular respiration: The processes by which cells generate ATP through glycolysis, the citric acid cycle, and oxidative phosphorylation.
-
Photosynthesis: How plants convert light energy into chemical energy in the form of ATP.
-
Muscle contraction: The role of ATP in providing energy for muscle fiber shortening.
-
Active transport: The movement of molecules against their concentration gradients, often powered by ATP hydrolysis.
-
Signal transduction: ATP hydrolysis and related phosphate transfer reactions play key roles in cellular signaling pathways.
-
DNA Replication and Repair: The energy for these critical processes is derived from ATP hydrolysis.
Conclusion
The hydrolysis of ATP is a cornerstone of cellular energetics. This reaction is decisively exergonic, releasing energy that fuels countless endergonic processes vital for life. While the standard free energy change provides a useful benchmark, the actual free energy change is modulated by various factors, including the concentrations of reactants and products, temperature, pH, and enzymatic activity. Understanding the thermodynamics and the mechanisms of ATP hydrolysis is paramount for a comprehensive understanding of life's intricate biochemical machinery. The exergonic nature of ATP hydrolysis allows it to act as the cell's primary energy currency, powering life's processes. Its study provides a gateway into the broader complexities of cellular energy transfer and metabolic pathways.
Latest Posts
Latest Posts
-
How To Choose U And Dv In Integration By Parts
Apr 03, 2025
-
Force On A Charge Moving In A Magnetic Field
Apr 03, 2025
-
Maximize The Area Of A Rectangle
Apr 03, 2025
-
What Is The Domain Of Tan
Apr 03, 2025
-
How To Find Ph Of A Salt
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Is The Hydrolysis Of Atp Endergonic Or Exergonic . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
