Is The Stationary Phase Polar Or Nonpolar
Muz Play
Apr 06, 2025 · 6 min read
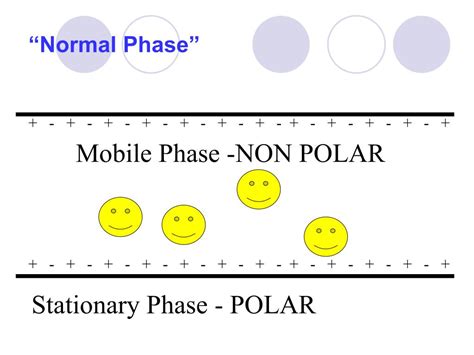
Table of Contents
Is the Stationary Phase Polar or Nonpolar? Understanding Chromatography
Chromatography, a cornerstone technique in analytical chemistry, relies heavily on the interaction between a stationary phase and the components of a sample mixture. A crucial aspect of understanding and optimizing chromatographic separations is the polarity of the stationary phase. This article delves deep into the question: Is the stationary phase polar or nonpolar? The answer, as we'll explore, isn't a simple yes or no, but depends entirely on the specific chromatographic technique being employed. We will examine various chromatographic methods, highlighting the nature of their stationary phases and how this impacts separation efficacy.
Understanding Polarity and its Role in Chromatography
Before diving into specific chromatographic techniques, let's establish a firm grasp on the concept of polarity. Polarity refers to the distribution of electrical charge within a molecule. Polar molecules possess a significant dipole moment due to an uneven distribution of electrons, leading to regions of partial positive and partial negative charge. Nonpolar molecules, conversely, exhibit a relatively even electron distribution, lacking a significant dipole moment.
In chromatography, the interaction between the stationary phase and the analyte (the substance being separated) is driven by intermolecular forces. These forces, including dipole-dipole interactions, hydrogen bonding, and van der Waals forces, are heavily influenced by the polarity of both the stationary phase and the analyte. Like dissolves like is a fundamental principle: polar analytes interact strongly with polar stationary phases, while nonpolar analytes prefer nonpolar stationary phases.
Common Chromatographic Techniques and their Stationary Phase Polarity
Several widely used chromatographic techniques employ different stationary phases with varying polarities. Understanding these differences is key to selecting the appropriate method for a specific separation challenge.
1. Gas Chromatography (GC)
Gas chromatography utilizes a gaseous mobile phase and typically employs a nonpolar stationary phase. Common stationary phases include:
-
Polydimethyl siloxane (PDMS): A highly nonpolar stationary phase, ideal for separating nonpolar and moderately polar compounds. Its inert nature minimizes analyte interactions beyond those governed by polarity.
-
Poly(5% diphenyl/95% dimethylsiloxane): This stationary phase offers a slightly increased polarity compared to pure PDMS, making it suitable for separating compounds with a broader range of polarities. The diphenyl groups introduce more polarizable sites.
The choice of stationary phase in GC is crucial for achieving optimal separation. A nonpolar stationary phase will retain nonpolar analytes longer, providing better separation from polar compounds that elute more quickly.
2. High-Performance Liquid Chromatography (HPLC)
HPLC boasts a much wider array of stationary phases, catering to a broad spectrum of analyte polarities. The stationary phase is typically packed into a column. Here are some common types:
-
Reversed-Phase HPLC: This is the most widely used mode in HPLC. It employs a nonpolar stationary phase (often a C18 alkyl chain bonded to silica gel) and a polar mobile phase (usually a mixture of water and organic solvents like acetonitrile or methanol). Nonpolar analytes interact strongly with the nonpolar stationary phase, resulting in longer retention times.
-
Normal-Phase HPLC: This method uses a polar stationary phase (like silica gel) and a nonpolar mobile phase (like hexane or heptane). Polar analytes interact strongly with the polar stationary phase, leading to longer retention times. This method is less common than reversed-phase HPLC due to the higher risk of peak tailing and the need for strictly anhydrous conditions.
-
Hydrophilic Interaction Chromatography (HILIC): HILIC uses a polar stationary phase (often a polar-modified silica gel) and a mobile phase containing a high percentage of organic solvent. This technique is particularly useful for separating polar and hydrophilic compounds, including sugars and peptides.
The choice between reversed-phase and normal-phase HPLC significantly impacts the separation outcome. Reversed-phase is preferred for most applications due to its robustness, versatility, and ease of use.
3. Thin-Layer Chromatography (TLC)
TLC is a simple and widely used technique for qualitative analysis and rapid separations. The stationary phase is a thin layer of adsorbent material, typically silica gel or alumina, coated on a glass or plastic plate.
-
Silica gel: This is the most common stationary phase in TLC and is polar in nature. Its surface contains silanol (Si-OH) groups, which can engage in hydrogen bonding and dipole-dipole interactions with polar analytes.
-
Alumina: Alumina is another common stationary phase in TLC, also considered polar. However, it's more strongly adsorbent than silica gel, making it suitable for separating compounds with a wide range of polarities.
TLC utilizes a non-polar mobile phase to separate compounds based on their differential interaction with the polar stationary phase. Polar compounds interact more strongly with the silica gel or alumina and move more slowly, while nonpolar compounds move more rapidly.
4. Supercritical Fluid Chromatography (SFC)
SFC bridges the gap between GC and HPLC. It uses a supercritical fluid (like carbon dioxide) as the mobile phase. The stationary phase can be either polar or nonpolar, depending on the specific application. Chiral stationary phases are also frequently used in SFC for enantiomer separation.
The flexibility in stationary phase selection allows SFC to separate a wide range of compounds, including those that are difficult to separate using either GC or HPLC.
Factors Influencing Stationary Phase Selection
The selection of the appropriate stationary phase is not solely determined by its polarity. Other factors play a crucial role:
-
Analyte properties: The polarity, size, and functionality of the analytes directly influence the choice of stationary phase.
-
Separation goals: The desired resolution (separation between peaks), speed of analysis, and sensitivity requirements all influence the selection process.
-
Mobile phase compatibility: The stationary phase must be compatible with the mobile phase used to avoid column degradation or unwanted interactions.
-
Column stability: The stability of the stationary phase under the chromatographic conditions is vital to ensure consistent and reliable results.
Optimizing Separations: Understanding the interplay between Stationary and Mobile Phases
The choice of stationary phase is only one piece of the puzzle in optimizing chromatographic separations. The mobile phase also plays a critical role. The mobile phase's polarity, strength, and composition significantly impact retention times and separation efficiency.
In reversed-phase HPLC, for example, the composition of the mobile phase (e.g., the percentage of water and organic solvent) is carefully controlled to adjust the retention times of the analytes. Increasing the percentage of organic solvent weakens the interaction between the nonpolar analyte and the stationary phase, leading to faster elution.
Similarly, in normal-phase HPLC, the strength of the mobile phase (e.g., the eluting power of the solvent) is adjusted to control the retention times. Stronger solvents elute analytes more rapidly.
Conclusion: A nuanced perspective on stationary phase polarity
In conclusion, the question of whether a stationary phase is polar or nonpolar is not a simple binary answer. The polarity of the stationary phase is highly dependent on the specific chromatographic technique employed. GC typically uses nonpolar stationary phases, while HPLC offers a broader range, including both polar and nonpolar options, depending on the mode of separation. TLC commonly uses polar stationary phases like silica gel. Understanding the interplay between stationary phase polarity, mobile phase composition, and analyte properties is crucial for achieving successful and efficient chromatographic separations. Careful consideration of these factors ensures optimal separation and accurate analytical results. This knowledge empowers scientists and researchers to tailor their chromatographic methods for specific applications, leading to improved analytical capabilities and better results. The ongoing development and refinement of new stationary phase materials continue to expand the capabilities of chromatography, pushing the boundaries of separation science and contributing to advancements in various fields.
Latest Posts
Latest Posts
-
What Is P Bar In Statistics
Apr 06, 2025
-
What Color Is The Cell Membrane
Apr 06, 2025
-
How To Solve Two Step Equations With Division
Apr 06, 2025
-
Density Is A Property Of Matter
Apr 06, 2025
-
The Most Likely Cause Of Bedding In This Image Is
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about Is The Stationary Phase Polar Or Nonpolar . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
