Molecules Consisting Only Of Carbon And Hydrogen Are Called
Muz Play
Apr 02, 2025 · 6 min read
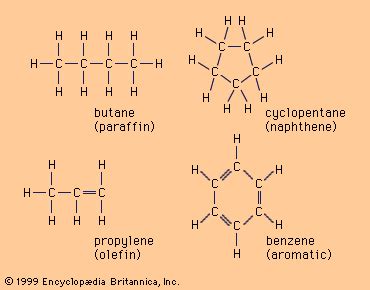
Table of Contents
Molecules Consisting Only of Carbon and Hydrogen are Called Hydrocarbons: A Deep Dive
Molecules consisting only of carbon and hydrogen are called hydrocarbons. These fundamental organic compounds form the basis of a vast array of natural and synthetic materials, impacting everything from the energy we consume to the plastics that shape our modern world. Understanding hydrocarbons, their diverse structures, properties, and applications, is crucial in numerous scientific disciplines, including chemistry, biology, geology, and engineering. This comprehensive guide will explore the fascinating world of hydrocarbons, examining their classification, nomenclature, properties, and significance.
What are Hydrocarbons?
Hydrocarbons are organic compounds composed exclusively of carbon (C) and hydrogen (H) atoms. The carbon atoms are linked together to form a carbon skeleton, with hydrogen atoms attached to satisfy the carbon's valence electrons. The simplicity of their composition belies the incredible diversity and complexity of hydrocarbon structures and the wide range of properties they exhibit. This diversity arises from the ability of carbon atoms to form long chains, branched chains, and ring structures.
Classification of Hydrocarbons: A Structural Overview
Hydrocarbons are broadly classified into two main categories based on the type of carbon-carbon bonds present:
1. Aliphatic Hydrocarbons: Chains and Branches
Aliphatic hydrocarbons feature carbon atoms arranged in open chains, which can be either straight chains or branched chains. This category further subdivides into:
-
Alkanes: These are saturated hydrocarbons, meaning all carbon-carbon bonds are single bonds. Each carbon atom is bonded to four other atoms (carbon or hydrogen), achieving a tetrahedral geometry. Alkanes are relatively unreactive due to the strong C-C and C-H bonds. Examples include methane (CH₄), ethane (C₂H₆), propane (C₃H₈), and butane (C₄H₁₀). The general formula for alkanes is C<sub>n</sub>H<sub>2n+2</sub>, where 'n' represents the number of carbon atoms.
-
Alkenes: These are unsaturated hydrocarbons containing at least one carbon-carbon double bond (C=C). The presence of the double bond introduces a region of higher electron density, making alkenes more reactive than alkanes. They readily undergo addition reactions. Examples include ethene (C₂H₄), propene (C₃H₆), and butene (C₄H₈). The general formula for alkenes is C<sub>n</sub>H<sub>2n</sub>.
-
Alkynes: These unsaturated hydrocarbons contain at least one carbon-carbon triple bond (C≡C). The triple bond represents a region of even higher electron density than the double bond in alkenes, making alkynes the most reactive of the aliphatic hydrocarbons. Examples include ethyne (C₂H₂), propyne (C₃H₄), and butyne (C₄H₆). The general formula for alkynes is C<sub>n</sub>H<sub>2n-2</sub>.
2. Aromatic Hydrocarbons: Rings of Resonance
Aromatic hydrocarbons are characterized by the presence of one or more benzene rings. A benzene ring consists of six carbon atoms arranged in a planar hexagonal structure with alternating single and double bonds. This arrangement creates a delocalized electron cloud above and below the plane of the ring, resulting in exceptional stability and unique chemical properties. Benzene (C₆H₆) is the simplest aromatic hydrocarbon. Other aromatic hydrocarbons include toluene (methylbenzene), xylene (dimethylbenzene), and naphthalene (two fused benzene rings).
Nomenclature of Hydrocarbons: Naming the Molecules
A systematic naming system, known as IUPAC nomenclature, is used to name hydrocarbons. This system ensures that each hydrocarbon has a unique and unambiguous name based on its structure. The rules involve identifying the longest carbon chain, numbering the carbons, naming the substituents (branches or functional groups), and arranging the names alphabetically.
Properties of Hydrocarbons: A Diverse Range
The properties of hydrocarbons vary significantly depending on their structure and molecular weight. Key properties include:
-
Physical State: Smaller hydrocarbons (e.g., methane, ethane, propane) are gases at room temperature, while larger hydrocarbons are liquids or solids.
-
Boiling Point: Boiling points generally increase with increasing molecular weight and chain length. Branched-chain hydrocarbons have lower boiling points than straight-chain hydrocarbons of the same molecular weight.
-
Solubility: Hydrocarbons are generally nonpolar and insoluble in water (a polar solvent). They are, however, soluble in nonpolar solvents such as other hydrocarbons.
-
Flammability: Hydrocarbons are highly flammable, reacting with oxygen to produce carbon dioxide, water, and heat. This flammability is the basis for their use as fuels.
-
Reactivity: Alkanes are relatively unreactive, while alkenes and alkynes are more reactive due to the presence of double and triple bonds respectively. Aromatic hydrocarbons exhibit a unique type of stability due to the delocalized electron system in the benzene ring.
Applications of Hydrocarbons: A World of Uses
Hydrocarbons have a wide range of applications, impacting various aspects of modern life:
1. Fuels: Powering Our World
Hydrocarbons are the primary source of energy for transportation, electricity generation, and heating. Natural gas (primarily methane), gasoline, diesel fuel, and kerosene are all mixtures of hydrocarbons derived from petroleum or natural gas.
2. Plastics and Polymers: Shaping Modern Life
Many plastics and polymers are derived from hydrocarbons. Polyethylene, polypropylene, and polystyrene are just a few examples of synthetic materials with widespread use in packaging, construction, and consumer goods.
3. Solvents: Dissolving Agents
Certain hydrocarbons, like hexane and heptane, are excellent solvents used in various industrial processes, including cleaning and extraction.
4. Pharmaceuticals and Other Chemicals: Building Blocks of Industry
Hydrocarbons serve as crucial building blocks in the synthesis of countless chemicals, including pharmaceuticals, pesticides, and other industrial chemicals.
5. Lubricants: Reducing Friction
Certain hydrocarbons are used as lubricants to reduce friction and wear in engines and machinery.
Environmental Concerns Related to Hydrocarbons: The Importance of Sustainability
While hydrocarbons are essential for modern life, their extraction, processing, and combustion contribute to several environmental concerns:
-
Greenhouse Gas Emissions: The combustion of hydrocarbons releases carbon dioxide (CO₂), a major greenhouse gas contributing to climate change.
-
Air Pollution: The incomplete combustion of hydrocarbons can produce harmful pollutants such as particulate matter and nitrogen oxides, impacting air quality.
-
Oil Spills: Accidents involving oil tankers or offshore drilling platforms can result in devastating oil spills, harming marine ecosystems.
-
Habitat Destruction: The extraction of hydrocarbons often involves significant habitat destruction and land disruption.
The Future of Hydrocarbons: Towards Sustainable Practices
The growing awareness of the environmental impact of hydrocarbon use is driving efforts to develop more sustainable practices. These include:
-
Development of Renewable Energy Sources: The transition towards renewable energy sources, such as solar, wind, and hydroelectric power, can reduce our reliance on hydrocarbon fuels.
-
Carbon Capture and Storage: Technologies are being developed to capture CO₂ emissions from hydrocarbon combustion and store them underground, preventing their release into the atmosphere.
-
Improved Efficiency and Conservation: Improving the efficiency of hydrocarbon use and promoting energy conservation can reduce overall consumption.
-
Development of Biofuels: Biofuels derived from renewable biomass sources can offer a more sustainable alternative to fossil fuels.
-
Circular Economy Approaches: Implementing circular economy principles, focusing on reducing waste and recycling materials, can minimize the environmental impact of hydrocarbon-based products.
Conclusion: Hydrocarbons - Essential but with Responsibility
Hydrocarbons are ubiquitous, impacting almost every aspect of our lives. Their versatility and energy density have propelled technological advancement, but their environmental impact necessitates a cautious and responsible approach. The future hinges on transitioning towards sustainable practices, embracing renewable energy sources, and developing innovative technologies to minimize the environmental footprint of hydrocarbons while leveraging their unique properties for critical applications. Understanding the structure, properties, and applications of hydrocarbons is paramount to fostering sustainable development and ensuring a healthier planet for generations to come.
Latest Posts
Latest Posts
-
How Does Molecular Biology Support The Theory Of Evolution
Apr 03, 2025
-
Not A And Not B Truth Table
Apr 03, 2025
-
What Is The Density Of Glass
Apr 03, 2025
-
Oxidation State Of O In H2o
Apr 03, 2025
-
Does Electron Withdrawing Groups Increase Acidity
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Molecules Consisting Only Of Carbon And Hydrogen Are Called . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
