To Increase The Concentration Of A Solution You Could
Muz Play
Apr 02, 2025 · 6 min read
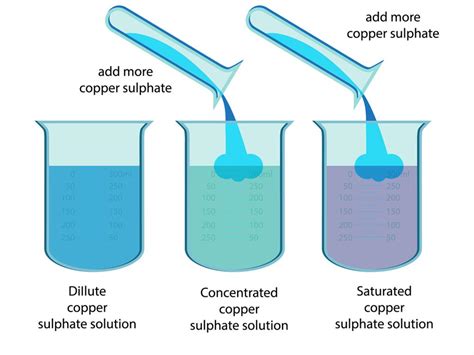
Table of Contents
To Increase the Concentration of a Solution: A Comprehensive Guide
Increasing the concentration of a solution is a fundamental process in chemistry and various other fields. Understanding the methods and implications is crucial for accurate results and safe practices. This comprehensive guide explores various techniques to achieve this, delving into the underlying principles and practical considerations.
Understanding Concentration and its Units
Before delving into the methods, let's clarify what we mean by concentration. Concentration refers to the amount of solute dissolved in a given amount of solvent or solution. It's expressed using various units, including:
- Molarity (M): Moles of solute per liter of solution. This is perhaps the most common unit used in chemistry.
- Molality (m): Moles of solute per kilogram of solvent. Molality is independent of temperature, unlike molarity.
- Normality (N): Equivalents of solute per liter of solution. This unit is used less frequently now.
- Percent concentration (%): Expressed as a percentage of solute by mass, volume, or mass/volume. For example, a 10% (w/v) solution contains 10 grams of solute per 100 mL of solution.
- Parts per million (ppm) and parts per billion (ppb): Used for extremely dilute solutions.
Methods to Increase Solution Concentration
Several methods can increase the concentration of a solution. The choice depends on the nature of the solute and solvent, the desired final concentration, and practical considerations.
1. Evaporation of Solvent
This is the simplest method, applicable to solutions where the solute is non-volatile and the solvent is volatile. By carefully evaporating the solvent, you increase the relative amount of solute, thus increasing the concentration.
How it Works: The solvent is removed, typically by heating the solution gently. This concentrates the remaining solute in the reduced volume of the solvent.
Considerations:
- Temperature Control: Overheating can damage the solute or lead to decomposition. Gentle heating and careful monitoring are crucial.
- Solvent Volatility: The method is only effective if the solvent is more volatile than the solute.
- Potential for Loss: Some solute may be lost during evaporation, especially if it's prone to sublimation or decomposition at higher temperatures.
- Crystallization: As the solvent evaporates, the solute may crystallize out of solution. This may be desirable or undesirable depending on the application.
2. Adding More Solute
The most straightforward method is to add more of the solute to the existing solution. This directly increases the amount of solute present, leading to a higher concentration.
How it Works: The solute is carefully added to the solution, ensuring thorough mixing to achieve uniform concentration.
Considerations:
- Solubility: The solute must be soluble in the solvent. Adding more solute beyond the solubility limit will result in undissolved solute remaining in the solution.
- Mixing: Thorough mixing is necessary to ensure a homogenous solution with uniform concentration.
- Saturation: It's important to be aware of the saturation point – the maximum amount of solute that can dissolve in a given amount of solvent at a specific temperature. Exceeding this point results in an unsaturated solution.
3. Using a Concentrated Stock Solution
Many laboratories and industries use stock solutions, which are highly concentrated solutions. Diluting a stock solution with a suitable solvent is a common way to prepare solutions of lower concentrations. However, to increase concentration, we can use the reverse process.
How it Works: A volume of concentrated stock solution is added to a smaller volume of a less concentrated solution or even just the solute itself. This increases the overall amount of solute in the final volume, resulting in a higher concentration.
Considerations:
- Accurate Measurements: Accurate measurement of both the stock solution and the solvent is critical for achieving the desired concentration.
- Mixing: Thorough mixing ensures uniformity.
- Safety: Handling concentrated solutions requires caution as they may be corrosive, toxic, or hazardous.
4. Fractional Crystallization/Precipitation
This technique is suitable when dealing with mixtures of solutes. By carefully manipulating the solubility of different components through changes in temperature or solvent, we can selectively crystallize or precipitate one component, leaving a more concentrated solution of the remaining components.
How it Works: Solubility changes with temperature. By cooling the solution slowly, the less soluble component may crystallize out. Alternatively, changes in solvent polarity may also be used to precipitate a solute.
Considerations:
- Solubility Data: Requires a good understanding of the solubility properties of the components involved.
- Purity: The crystallized or precipitated solute may not be perfectly pure and may require further purification steps.
- Yield: The yield of the desired concentrated solution may be affected by various factors like temperature control, rate of cooling, and solvent choice.
5. Reverse Osmosis
This is a membrane-based separation technique that utilizes pressure to force solvent molecules through a semi-permeable membrane, leaving behind the solute. This is primarily used for water purification but can be adapted for concentrating dissolved substances.
How it Works: A pressure greater than the osmotic pressure is applied to the solution. This forces the solvent through the membrane, leaving a more concentrated solution behind.
Considerations:
- Membrane Selection: The membrane's pore size and material must be chosen carefully based on the solute and solvent.
- Pressure: High pressure is required, necessitating specialized equipment.
- Membrane Fouling: The membrane may become fouled over time, reducing its efficiency.
Calculating the Final Concentration
Accurately calculating the final concentration after employing any of the above methods is essential. This typically involves using the appropriate concentration units and applying dilution or concentration formulas.
For example, the dilution formula, M1V1 = M2V2 (where M is molarity and V is volume), can be rearranged to calculate the final concentration after adding more solvent. Conversely, to calculate the concentration after adding more solute, one needs to consider the total moles of solute and total volume of the solution.
Example: Let's say you have 100 mL of a 0.5 M solution, and you want to increase its concentration to 1 M by adding more solute. You need to determine how much solute to add. This would involve calculations considering the molar mass of the solute and its solubility.
Safety Precautions
When working with solutions and chemicals, always prioritize safety:
- Wear appropriate personal protective equipment (PPE): This includes safety goggles, gloves, and lab coats.
- Work in a well-ventilated area: Some solvents and solutes can be toxic or have harmful fumes.
- Handle chemicals with care: Avoid contact with skin and eyes.
- Dispose of waste properly: Follow appropriate waste disposal procedures.
Conclusion
Increasing the concentration of a solution involves several techniques, each with its own advantages and limitations. Choosing the optimal method depends on factors like the type of solute and solvent, the desired concentration, and available resources. Understanding the principles involved, performing accurate calculations, and strictly adhering to safety protocols are crucial for successful concentration and handling of chemical solutions. Always prioritize safety and accurate measurements to avoid errors and ensure reliable results. Remember that the best method will depend on your specific application and resources available. Consult relevant literature and safety data sheets for guidance on specific chemicals and procedures.
Latest Posts
Latest Posts
-
Similarities Between Endocrine And Nervous System
Apr 03, 2025
-
How To Choose U And Dv In Integration By Parts
Apr 03, 2025
-
Force On A Charge Moving In A Magnetic Field
Apr 03, 2025
-
Maximize The Area Of A Rectangle
Apr 03, 2025
-
What Is The Domain Of Tan
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about To Increase The Concentration Of A Solution You Could . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
