What Are The Characteristics Of Enzymes
Muz Play
Apr 02, 2025 · 7 min read
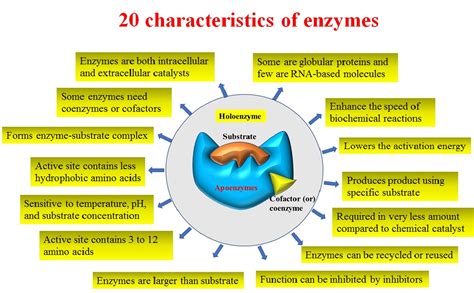
Table of Contents
What are the Characteristics of Enzymes?
Enzymes are biological catalysts, meaning they speed up chemical reactions within living organisms without being consumed in the process. Understanding their characteristics is crucial to comprehending the intricate workings of life itself. These remarkable molecules exhibit a range of properties that dictate their function and effectiveness, making them indispensable for virtually every biological process. This article delves deep into the key characteristics of enzymes, exploring their structure, function, and the factors influencing their activity.
I. The Nature of Enzyme Catalysis
Before diving into specific characteristics, let's establish a foundation. Enzymes achieve catalysis by lowering the activation energy of a reaction. This means they reduce the energy barrier required for a reaction to proceed, thus accelerating the rate at which reactants are converted into products. This isn't achieved by altering the overall energy change (ΔG) of the reaction – the difference in energy between reactants and products remains the same – but by providing an alternative, lower-energy pathway. This happens through the formation of a temporary complex between the enzyme and its substrate(s), the molecules upon which the enzyme acts.
A. Substrate Specificity: The Lock and Key Model (and its refinements)
One of the most defining characteristics of enzymes is their specificity. This refers to their ability to selectively bind to specific substrate molecules and catalyze only certain reactions. The classic explanation for this specificity is the lock-and-key model, where the enzyme's active site (the region where the substrate binds) possesses a rigid, complementary shape to the substrate. Only the correct "key" (substrate) will fit into the "lock" (enzyme).
However, this model is an oversimplification. The more accurate induced-fit model proposes that the enzyme's active site is flexible and undergoes conformational changes upon substrate binding. This interaction induces a change in the enzyme's shape, optimizing the fit and facilitating catalysis. This dynamic interaction enhances the enzyme's ability to bind to and process its substrates efficiently.
II. Key Characteristics of Enzymes
Let's now delve into the core characteristics that define enzymes:
A. Protein Nature (Mostly): The Building Blocks of Enzymes
The vast majority of enzymes are proteins. Their complex three-dimensional structures, determined by their amino acid sequences, create specific active sites capable of binding substrates and catalyzing reactions. The precise arrangement of amino acids within the active site dictates the enzyme's specificity and catalytic mechanism. However, a small number of catalytic RNA molecules, called ribozymes, also exhibit enzymatic activity.
B. Catalytic Efficiency: Accelerating Reaction Rates Dramatically
Enzymes are incredibly efficient catalysts, often accelerating reaction rates by factors of millions or even billions compared to uncatalyzed reactions. This phenomenal efficiency is a direct consequence of their ability to lower activation energy and precisely orient substrates within the active site.
C. Mild Reaction Conditions: Operating Under Physiological Parameters
Enzymes function effectively under relatively mild conditions of temperature, pH, and pressure that are compatible with life. This contrasts sharply with many chemical catalysts, which often require harsh conditions (high temperature, extreme pH) to function. The ability to operate under physiological conditions is crucial for maintaining cellular integrity and preventing damage to biological molecules.
D. Regulation: Controlling Enzymatic Activity
Enzymes are not always "on." Their activity is often regulated to meet the changing metabolic demands of the cell. This regulation can be achieved through various mechanisms, including:
- Allosteric regulation: Binding of molecules (allosteric effectors) to sites other than the active site can either activate or inhibit enzyme activity.
- Covalent modification: Chemical modifications such as phosphorylation or glycosylation can alter enzyme activity.
- Feedback inhibition: The end product of a metabolic pathway inhibits an earlier enzyme in the pathway, preventing overproduction.
- Enzyme concentration: The amount of enzyme present in a cell can be controlled through gene expression.
E. Sensitivity to Environmental Factors: The Influence of External Conditions
Enzyme activity is highly sensitive to changes in its environment. Several factors can significantly affect their function:
-
Temperature: Enzymes have an optimal temperature at which they function most efficiently. Higher temperatures can denature the protein, disrupting its three-dimensional structure and rendering it inactive. Conversely, lower temperatures can slow down the reaction rate.
-
pH: Each enzyme has an optimal pH range. Deviation from this range can alter the charge distribution in the active site, affecting substrate binding and catalysis. Extreme pH values can also denature the enzyme.
-
Substrate concentration: The rate of an enzyme-catalyzed reaction increases with increasing substrate concentration up to a certain point (saturation). At saturation, all active sites are occupied, and further increases in substrate concentration do not increase the reaction rate.
-
Enzyme concentration: The rate of an enzyme-catalyzed reaction is directly proportional to the enzyme concentration, provided that there is sufficient substrate. Increasing enzyme concentration increases the number of active sites available, leading to a faster reaction rate.
-
Inhibitors: Certain molecules, called inhibitors, can bind to enzymes and reduce or completely block their activity. Inhibitors can be competitive (competing with the substrate for the active site) or non-competitive (binding to a different site and altering the enzyme's shape).
-
Activators: Conversely, some molecules can activate enzymes, enhancing their catalytic activity.
F. Specificity of the Active Site: A Precise Molecular Interaction
The active site is a crucial region within the enzyme's three-dimensional structure. It is characterized by its high specificity for the substrate. This specificity arises from the precise arrangement of amino acid side chains within the active site, which interact with the substrate through various non-covalent forces (hydrogen bonds, ionic interactions, van der Waals forces, hydrophobic interactions). The unique architecture of the active site ensures that only the correct substrate can bind and undergo catalysis.
G. Turnover Number: A Measure of Catalytic Efficiency
The turnover number (also known as kcat) is a measure of an enzyme's catalytic efficiency. It represents the number of substrate molecules converted to product per enzyme molecule per unit time (e.g., per second) when the enzyme is saturated with substrate. A higher turnover number indicates a more efficient enzyme.
H. Cofactors and Coenzymes: Essential Helpers
Many enzymes require non-protein components, called cofactors, for their activity. These cofactors can be metal ions (e.g., Mg2+, Zn2+) or organic molecules called coenzymes. Cofactors often participate directly in the catalytic mechanism, assisting in substrate binding or facilitating chemical transformations. Many vitamins serve as precursors to coenzymes.
I. Isozymes: Enzyme Variants with Similar Functions
Isozymes are different forms of the same enzyme that catalyze the same reaction but may have slightly different properties, such as optimal pH or substrate affinity. Isozymes often exhibit tissue-specific expression, meaning different isozymes may be found in different tissues or organs. This allows for fine-tuning of enzyme activity to meet the specific metabolic needs of different tissues.
III. The Importance of Studying Enzyme Characteristics
Understanding the characteristics of enzymes is paramount for numerous reasons:
-
Medical applications: Enzymes are crucial targets for drug development. Inhibitors of specific enzymes are used to treat various diseases, including infections, cancer, and metabolic disorders.
-
Industrial applications: Enzymes are used extensively in various industries, including food processing, textile manufacturing, and biofuel production. Their ability to catalyze reactions under mild conditions makes them environmentally friendly alternatives to traditional chemical catalysts.
-
Agricultural applications: Enzymes are used in agriculture to enhance crop yields, improve nutrient utilization, and develop pest-resistant crops.
-
Research applications: Enzymes are essential tools in molecular biology and biochemistry research. They are used in various techniques, such as PCR, DNA sequencing, and protein purification.
-
Environmental applications: Enzymes are employed in bioremediation, the use of biological agents to clean up environmental pollutants.
IV. Conclusion: Enzymes – The Workhorses of Life
Enzymes are truly remarkable molecules. Their unique characteristics – catalytic efficiency, specificity, sensitivity to environmental factors, and regulation – make them essential for life. Understanding their properties provides valuable insights into the complex mechanisms that govern biological processes and offers opportunities to harness their power for diverse applications in medicine, industry, agriculture, and environmental science. Further research into enzyme structure and function continues to unlock new possibilities for their utilization and manipulation, driving advances across a wide spectrum of scientific disciplines.
Latest Posts
Latest Posts
-
Where Are The Cell Bodies For The Sensory Neurons Located
Apr 03, 2025
-
Difference Between Amorphous Solid And Crystalline Solid
Apr 03, 2025
-
Alkali Metals Alkaline Earth Metals Halogens And Noble Gases
Apr 03, 2025
-
What Determines Primary Structure Of A Protein
Apr 03, 2025
-
Chromosomes Disperse And Are No Longer Visible
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about What Are The Characteristics Of Enzymes . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
