What Is The Five-carbon Sugar Found In Dna.
Muz Play
Apr 05, 2025 · 6 min read
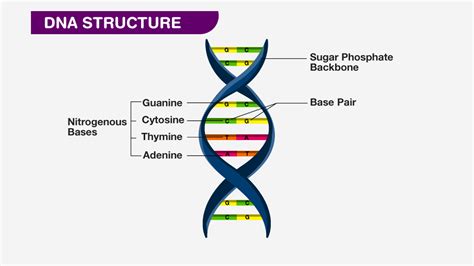
Table of Contents
What is the Five-Carbon Sugar Found in DNA?
Deoxyribose: The Backbone of Life's Blueprint
DNA, or deoxyribonucleic acid, is the fundamental molecule of heredity. It carries the genetic instructions for the development, functioning, growth, and reproduction of all known organisms and many viruses. At the heart of this remarkable molecule lies a crucial component: a five-carbon sugar known as deoxyribose. Understanding deoxyribose's structure and function is key to understanding DNA itself. This article delves deep into the characteristics of deoxyribose, exploring its chemical structure, its role in DNA's stability and function, and its contrast with ribose, the sugar found in RNA.
The Chemical Structure of Deoxyribose
Deoxyribose is a pentose sugar, meaning it's a monosaccharide (simple sugar) with five carbon atoms. Its chemical formula is C₅H₁₀O₄. Unlike many other sugars, deoxyribose exists primarily in a cyclic form, a five-membered ring structure. This ring is composed of four carbon atoms and one oxygen atom. The fifth carbon atom is attached to the ring's oxygen atom, extending outwards.
Distinguishing Deoxyribose from Ribose
The key difference between deoxyribose and ribose, the sugar found in RNA (ribonucleic acid), lies in the presence or absence of a hydroxyl (-OH) group on the 2' carbon atom. Ribose possesses a hydroxyl group at this position, while deoxyribose lacks it. This seemingly minor difference has profound consequences for the properties and functions of DNA and RNA. The absence of the hydroxyl group in deoxyribose makes DNA more stable and less susceptible to hydrolysis (breakdown by water) compared to RNA.
The Significance of the 2' Carbon
The 2' carbon's hydroxyl group in ribose makes RNA less stable than DNA. This instability is linked to the increased reactivity of the hydroxyl group, which makes RNA more prone to hydrolysis. This inherent instability is partially responsible for RNA's role as a relatively short-lived intermediary molecule in protein synthesis, while DNA, with its greater stability, serves as the long-term storage repository for genetic information.
Deoxyribose's Role in DNA Structure
Deoxyribose forms the backbone of the DNA molecule. Each deoxyribose molecule is linked to a phosphate group and a nitrogenous base, forming a nucleotide. The nucleotides are linked together to form the polynucleotide chains of DNA.
The Phosphodiester Bond
The key linkage between the deoxyribose sugars in the DNA backbone is a phosphodiester bond. This strong covalent bond connects the 3' carbon of one deoxyribose molecule to the 5' carbon of the next deoxyribose molecule through a phosphate group. This creates a sugar-phosphate backbone that runs continuously along the length of the DNA molecule.
The 3' and 5' Ends
The directional nature of the phosphodiester bond gives DNA strands a defined polarity. One end of the DNA strand has a free 3' hydroxyl group, while the other end has a free 5' phosphate group. This polarity is crucial for DNA replication and transcription, as enzymes involved in these processes interact with the DNA strand in a specific direction.
Deoxyribose and DNA Stability
The lack of the 2' hydroxyl group in deoxyribose contributes significantly to DNA's remarkable stability. Several factors contribute to this stability:
Resistance to Hydrolysis
As mentioned, the absence of the 2' hydroxyl group makes DNA less susceptible to hydrolysis compared to RNA. This is because the hydroxyl group in ribose can participate in reactions that break the sugar-phosphate backbone. The greater stability of DNA is essential for maintaining the integrity of the genetic information it carries over long periods.
Double Helix Formation
The specific configuration of deoxyribose within the DNA molecule contributes to the formation of the iconic double helix structure. This double helix structure, with its two antiparallel strands wound around each other, provides additional stability to the DNA molecule. The bases on the inside of the helix interact through hydrogen bonds, further stabilizing the structure.
DNA Packaging
The double helix structure, made possible by deoxyribose's properties, allows for highly efficient packaging of DNA within the cell. DNA is packaged with histone proteins, forming chromatin fibers, and further compacted into chromosomes, which facilitates its organization and protection.
Deoxyribose and DNA Function
The structural features of deoxyribose are intimately linked to DNA's ability to function as the carrier of genetic information:
DNA Replication
During DNA replication, the double helix unwinds, and each strand serves as a template for the synthesis of a new complementary strand. The precise structure of deoxyribose and its role in the phosphodiester backbone allow for accurate replication of the genetic information. Enzymes such as DNA polymerase utilize the existing strand as a template, adding nucleotides in the 5' to 3' direction, guided by the complementary base pairing rules.
DNA Transcription
DNA transcription is the process of copying the genetic information from DNA to RNA. The structure of deoxyribose within the DNA molecule allows for the binding of RNA polymerase and other transcription factors. The precise unwinding and rewinding of the DNA double helix during transcription is dependent on the integrity of the deoxyribose-phosphate backbone.
DNA Repair
DNA is constantly exposed to factors that can cause damage, such as radiation and chemical mutagens. Cells possess sophisticated DNA repair mechanisms to maintain the integrity of their genetic information. These repair mechanisms often involve recognition and excision of damaged DNA segments, followed by resynthesis guided by the undamaged complementary strand. Deoxyribose's contribution to structural stability is crucial for the efficiency of these repair processes.
Deoxyribose: A Cornerstone of Life
In conclusion, deoxyribose is far more than just a simple sugar; it's a critical component of the DNA molecule, influencing its stability, structure, and function. The absence of the 2' hydroxyl group, differentiating it from ribose, is a key factor contributing to DNA's remarkable stability, allowing for the accurate storage and transmission of genetic information across generations. Its role in the phosphodiester backbone dictates the directionality and overall architecture of DNA, enabling processes like replication, transcription, and repair, which are essential for life as we know it. The seemingly simple pentose sugar, deoxyribose, is therefore a cornerstone of life's blueprint, a testament to the elegant simplicity and powerful functionality inherent in the molecules that underpin all living things. Further research into deoxyribose and its interactions within the larger DNA molecule continues to unlock deeper understanding of life’s intricate processes and the fundamental mechanisms of heredity. Its significance extends beyond the basic understanding of DNA structure, impacting fields such as medicine, biotechnology, and evolutionary biology. The study of this seemingly simple sugar provides invaluable insight into the complexity and beauty of life itself.
Latest Posts
Latest Posts
-
What Is The Main Transformation That Occurs During Glycolysis
Apr 05, 2025
-
Why Does The Atomic Radii Increase Down A Group
Apr 05, 2025
-
Valves That Prevent Backflow Of Blood Into The Ventricles
Apr 05, 2025
-
What Is The Mass Of Neutron In Amu
Apr 05, 2025
-
What Causes Semilunar Valves To Open
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about What Is The Five-carbon Sugar Found In Dna. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
