Where Is Oxide On The Periodic Table
Muz Play
Apr 02, 2025 · 6 min read
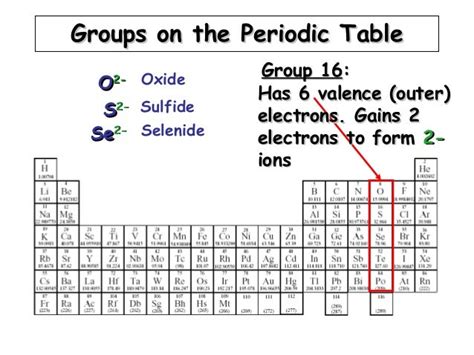
Table of Contents
Where is Oxide on the Periodic Table? Understanding Oxides and their Formation
The question "Where is oxide on the periodic table?" is a bit of a trick question. Oxide isn't an element, and therefore doesn't have a specific location on the periodic table. Instead, "oxide" refers to a compound containing at least one oxygen anion (O²⁻) and one other element's cation. Understanding where oxides appear on the periodic table requires exploring the reactivity of oxygen and its tendency to form compounds with a vast range of elements.
This comprehensive guide will delve into the fascinating world of oxides, explaining how they are formed, their properties, and their ubiquitous presence in our world, all while clarifying why you won't find "oxide" itself on Mendeleev's masterpiece.
Oxygen's Position and Reactivity: The Key to Oxide Formation
Oxygen, denoted by the symbol O and atomic number 8, sits in Group 16 (also known as Group VIA or the chalcogens) of the periodic table. Its position is crucial in understanding its remarkable tendency to form oxides. Oxygen's electronic configuration ([He] 2s²2p⁴) means it needs two more electrons to achieve a stable octet, mirroring the noble gas neon. This high electronegativity makes it a highly reactive element, readily accepting electrons from other atoms to form negatively charged oxide ions (O²⁻).
Types of Oxides: A Diverse Chemical Family
The properties of oxides vary drastically depending on the element they are combined with. This diversity arises from the differing electronegativities and oxidation states of the elements involved. We can broadly categorize oxides into several key types:
1. Basic Oxides: Reactions with Acids
Basic oxides are typically formed by the highly electropositive metals located on the left side of the periodic table, including Groups 1 and 2 (alkali and alkaline earth metals). These metals readily lose electrons to oxygen, forming ionic oxides with a predominantly ionic bond character. A classic example is sodium oxide (Na₂O), where sodium atoms donate electrons to oxygen atoms to form Na⁺ and O²⁻ ions. These oxides react readily with acids, forming salts and water:
- Example: Na₂O + 2HCl → 2NaCl + H₂O
The basicity of these oxides generally increases down a group and decreases across a period.
2. Acidic Oxides: Reactions with Bases
Acidic oxides are usually formed by nonmetals and some metalloids located on the right side of the periodic table. These oxides react with water to form acids or with bases to form salts and water. A prime example is sulfur trioxide (SO₃), which reacts with water to form sulfuric acid (H₂SO₄), a strong acid:
- Example: SO₃ + H₂O → H₂SO₄
The acidity of oxides generally increases across a period and decreases down a group.
3. Amphoteric Oxides: Dual Reactivity
Amphoteric oxides exhibit a dual nature, exhibiting both acidic and basic properties. They can react with both acids and bases, forming salts and water in both cases. Aluminum oxide (Al₂O₃) is a classic example:
- Reaction with acid: Al₂O₃ + 6HCl → 2AlCl₃ + 3H₂O
- Reaction with base: Al₂O₃ + 2NaOH + 3H₂O → 2Na[Al(OH)₄] (Sodium aluminate)
The amphoteric behavior often reflects the intermediate electronegativity of the metal involved. Transition metal oxides frequently display amphoteric character.
4. Neutral Oxides: Limited Reactivity
Neutral oxides display minimal reactivity with either acids or bases. Carbon monoxide (CO) and nitrous oxide (N₂O) are prime examples of neutral oxides. Their inertness stems from strong covalent bonding within the molecule and the stable oxidation states of the constituent elements.
5. Peroxides and Superoxides: Oxygen-Oxygen Bonds
Peroxides contain the peroxide ion (O₂²⁻), which has an oxygen-oxygen single bond. Hydrogen peroxide (H₂O₂) is a common example. Superoxides, containing the superoxide ion (O₂⁻), have a weaker oxygen-oxygen bond. Potassium superoxide (KO₂) is an example and is used in some breathing apparatus. These compounds show different reactivity patterns than typical oxides.
Mapping Oxides on the Periodic Table: Trends and Patterns
While "oxide" itself isn't on the periodic table, we can map the formation of oxides across it. Several key trends and patterns emerge:
-
Across a Period: Moving from left to right, the oxides become increasingly acidic. This is because the electronegativity of the element increases, leading to a greater polarization of the oxygen-element bond and a stronger tendency to form acidic oxides.
-
Down a Group: Moving down a group, the oxides of metals become increasingly basic, while the oxides of nonmetals become increasingly less acidic. This reflects changes in electronegativity and ionic radii down a group.
-
Transition Metals: Transition metals often exhibit multiple oxidation states, resulting in a variety of oxides with varying properties. For example, iron can form both FeO (iron(II) oxide) and Fe₂O₃ (iron(III) oxide), each displaying different properties and reactivities.
-
Metalloids: Metalloids tend to form amphoteric oxides, reflecting their intermediate position between metals and nonmetals.
Practical Applications of Oxides: A World of Oxides
Oxides are ubiquitous in the natural world and have countless applications in various industries:
-
Metallurgy: Many metal ores are oxides, which are processed to extract the pure metals. The reduction of metal oxides is a fundamental process in metallurgy.
-
Construction Materials: Oxides are used extensively in construction. Cement, for instance, is based on calcium oxide (CaO), while silicon dioxide (SiO₂) is a key component of sand and glass.
-
Catalysis: Many oxides act as catalysts in chemical reactions, influencing reaction rates without being consumed themselves. Vanadium oxide (V₂O₅) is a crucial catalyst in the production of sulfuric acid.
-
Electronics: Metal oxides are used extensively in electronic components, such as transistors and integrated circuits. Titanium dioxide (TiO₂) is used in many paints and sunscreens.
-
Medicine: Some metal oxides are used in medicine, for example, zinc oxide (ZnO) is used in topical ointments.
Beyond Simple Oxides: Complex Oxide Structures
The world of oxides extends far beyond simple binary compounds. Many complex oxides exist, featuring multiple metal cations and intricate crystal structures. These complex oxides often exhibit unique and valuable properties not found in simpler oxides. Examples include perovskites, spinels, and other complex structures often used in advanced materials applications like superconductors and catalysts.
Conclusion: Understanding the Chemistry of Oxides
While you won't find "oxide" as a single element on the periodic table, understanding oxygen's position and reactivity helps explain the vast array of oxides and their properties. The location of an element on the periodic table provides critical information about its electronegativity and its ability to form bonds with oxygen, ultimately determining the characteristics of the resulting oxide. From basic to acidic, amphoteric to neutral, oxides play a fundamental role in chemistry and have myriad applications across numerous fields. This expansive understanding of oxides underscores their significance and the crucial role of the periodic table in comprehending their diverse nature and widespread applications.
Latest Posts
Latest Posts
-
Similarities Between Endocrine And Nervous System
Apr 03, 2025
-
How To Choose U And Dv In Integration By Parts
Apr 03, 2025
-
Force On A Charge Moving In A Magnetic Field
Apr 03, 2025
-
Maximize The Area Of A Rectangle
Apr 03, 2025
-
What Is The Domain Of Tan
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Where Is Oxide On The Periodic Table . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
