Which Part Of Amino Acid Is Always Acidic
Muz Play
Apr 02, 2025 · 5 min read
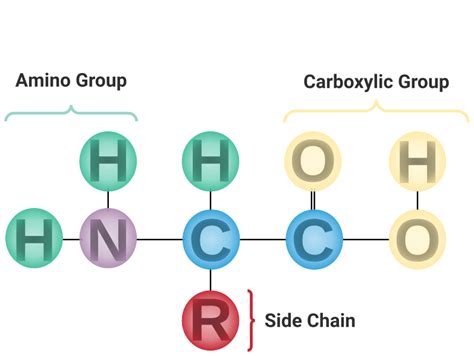
Table of Contents
Which Part of an Amino Acid is Always Acidic?
The defining characteristic of an amino acid is its possession of both an amino group (-NH2) and a carboxyl group (-COOH). While the amino group is basic, it is the carboxyl group that is always acidic. This article will delve deep into why this is the case, exploring the chemical properties of the carboxyl group, its behavior in aqueous solutions, and its implications for amino acid structure and function. We will also examine exceptions and nuances to this rule, ensuring a comprehensive understanding of amino acid acidity.
The Carboxyl Group: A Master of Acid-Base Chemistry
The carboxyl group, a combination of a carbonyl group (C=O) and a hydroxyl group (-OH), is the key player in the acidic nature of amino acids. Its acidity stems from the unique properties of this functional group.
Understanding Acidity: Proton Donation
Acidity is a measure of a substance's ability to donate a proton (H+). A strong acid readily donates its proton, while a weak acid donates it less readily. The carboxyl group's acidity arises from the ability of its hydroxyl group to release a proton. This is due to several factors:
-
Resonance Stabilization: After the proton is released, the negative charge on the remaining carboxylate ion (-COO-) is delocalized across the two oxygen atoms through resonance. This delocalization stabilizes the negative charge, making it easier for the proton to depart. The resonance structures distribute the negative charge, preventing it from being concentrated on a single atom, leading to greater stability. This stabilization significantly lowers the energy of the carboxylate ion, increasing the likelihood of proton donation.
-
Electronegativity: Oxygen is highly electronegative, meaning it attracts electrons strongly. This pulls electron density away from the O-H bond, weakening the bond and making it easier to break and release the proton. The electronegative oxygen atoms effectively stabilize the negative charge that develops on the carboxylate ion after proton dissociation.
-
Inductive Effect: The carbonyl group (C=O) exerts an inductive effect, further withdrawing electron density from the O-H bond. This effect enhances the electron-withdrawing power of the oxygen atom, facilitating proton release.
These combined factors—resonance stabilization, electronegativity, and inductive effect—work synergistically to make the carboxyl group a relatively strong acid compared to other functional groups typically found in organic molecules.
pKa and Acid Dissociation
The strength of an acid is often quantified using its pKa value. The pKa represents the pH at which half of the acid molecules have donated their proton. A lower pKa value indicates a stronger acid. The carboxyl group of amino acids typically has a pKa value in the range of 2-3. This means that at physiological pH (around 7.4), the carboxyl group will be largely deprotonated, existing as a carboxylate ion (-COO-).
Amino Acid Structure and the Carboxyl Group's Role
The carboxyl group's acidic nature is fundamental to the structure and function of amino acids.
Zwitterionic Form
At neutral pH, amino acids exist predominantly as zwitterions. A zwitterion is a molecule with both a positive and a negative charge. In amino acids, the amino group (-NH2) accepts a proton to become positively charged (-NH3+), while the carboxyl group donates a proton to become negatively charged (-COO-). This zwitterionic form is crucial for the amphoteric nature of amino acids—their ability to act as both acids and bases.
Peptide Bond Formation
The carboxyl group's acidity is essential for the formation of peptide bonds. Peptide bonds link amino acids together to form proteins. The reaction involves the carboxyl group of one amino acid reacting with the amino group of another amino acid, releasing a water molecule. This reaction requires the carboxyl group to be in its deprotonated form (-COO-) to facilitate the nucleophilic attack by the amino group. The acidic nature of the carboxyl group makes this reaction possible.
Interactions with Other Molecules
The carboxyl group’s charge and ability to donate or accept protons enables it to participate in various interactions with other molecules. This is crucial for protein folding, enzyme-substrate interactions, and other biological processes. The charged carboxylate group can engage in ionic interactions, hydrogen bonds, and other non-covalent interactions vital for protein structure and function.
Exceptions and Nuances
While the carboxyl group is always acidic in amino acids, the exact degree of acidity can vary slightly depending on the amino acid's side chain (R group). The side chain can influence the electron density around the carboxyl group through inductive effects or steric hindrance. However, these effects are usually minor and do not negate the fundamental acidity of the carboxyl group.
For example, some amino acids with electron-withdrawing groups in their side chains may have slightly lower pKa values for their carboxyl groups. Conversely, amino acids with electron-donating groups might exhibit slightly higher pKa values. However, in all cases, the carboxyl group remains the acidic part of the amino acid molecule.
Conclusion: The Unwavering Acidity of the Carboxyl Group
In conclusion, the carboxyl group (-COOH) is the unequivocally acidic component of an amino acid. Its acidity, stemming from the combined effects of resonance stabilization, electronegativity of oxygen, and the inductive effect of the carbonyl group, is central to amino acid properties. It dictates their behavior in solution, facilitates peptide bond formation, and contributes significantly to their roles in biological processes. Although minor variations in pKa values can be observed due to side-chain influence, the fundamental acidic nature of the carboxyl group remains a defining characteristic of all amino acids. Understanding this inherent acidity is paramount for comprehending protein structure, function, and the intricacies of biochemistry.
Latest Posts
Latest Posts
-
Similarities Between Endocrine And Nervous System
Apr 03, 2025
-
How To Choose U And Dv In Integration By Parts
Apr 03, 2025
-
Force On A Charge Moving In A Magnetic Field
Apr 03, 2025
-
Maximize The Area Of A Rectangle
Apr 03, 2025
-
What Is The Domain Of Tan
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Which Part Of Amino Acid Is Always Acidic . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
