Why Do Atoms Form Bonds With Other Atoms
Muz Play
Mar 31, 2025 · 7 min read
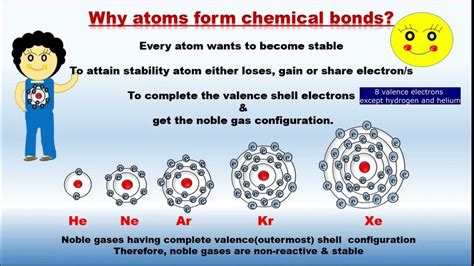
Table of Contents
- Why Do Atoms Form Bonds With Other Atoms
- Table of Contents
- Why Do Atoms Form Bonds with Other Atoms? A Deep Dive into Chemical Bonding
- The Drive for Stability: Octet Rule and Electron Configuration
- Understanding Electron Shells and Orbitals
- Types of Chemical Bonds: A Detailed Exploration
- 1. Ionic Bonds: The Dance of Ions
- 2. Covalent Bonds: Sharing is Caring
- 3. Metallic Bonds: A Sea of Electrons
- Beyond the Basics: Factors Influencing Bond Formation
- The Importance of Chemical Bonding in the World Around Us
- Conclusion: A Continuous Quest for Stability
- Latest Posts
- Latest Posts
- Related Post
Why Do Atoms Form Bonds with Other Atoms? A Deep Dive into Chemical Bonding
Atoms, the fundamental building blocks of matter, don't typically exist in isolation. Instead, they have a remarkable tendency to interact and bond with other atoms, forming molecules and complex structures that make up everything we see around us. But why? This seemingly simple question opens the door to a fascinating exploration of chemical bonding, electron configurations, and the very nature of stability in the universe.
The Drive for Stability: Octet Rule and Electron Configuration
At the heart of atomic bonding lies the quest for stability. Atoms strive to achieve a stable electron configuration, usually resembling that of a noble gas. This is famously known as the octet rule, which states that atoms tend to gain, lose, or share electrons to achieve eight electrons in their outermost shell, also known as the valence shell. Exceptions exist, particularly with elements having fewer than eight valence electrons, but the octet rule provides a useful framework for understanding bonding.
Understanding Electron Shells and Orbitals
To fully grasp why atoms bond, we need to delve into the structure of an atom. Electrons occupy specific energy levels, or shells, surrounding the nucleus. These shells are further subdivided into orbitals, which describe the probability of finding an electron in a particular region of space. Each orbital can hold a maximum of two electrons.
The outermost shell, the valence shell, plays a crucial role in bonding. Atoms with a nearly full valence shell tend to gain electrons, while those with a nearly empty valence shell tend to lose electrons. Atoms with a partially filled valence shell often share electrons to achieve stability.
Types of Chemical Bonds: A Detailed Exploration
There are several ways atoms can achieve a stable electron configuration and achieve this goal: through ionic bonds, covalent bonds, and metallic bonds. Each type has its unique characteristics and influences the properties of the resulting substance.
1. Ionic Bonds: The Dance of Ions
Ionic bonds form when one atom transfers one or more electrons to another atom. This transfer creates two oppositely charged ions: a positively charged cation (the atom that lost electrons) and a negatively charged anion (the atom that gained electrons). The electrostatic attraction between these oppositely charged ions holds them together in an ionic bond.
Examples: Sodium chloride (NaCl), or common table salt, is a classic example. Sodium (Na) readily loses one electron to achieve a stable configuration, becoming a Na⁺ ion. Chlorine (Cl) readily gains one electron, becoming a Cl⁻ ion. The strong electrostatic attraction between Na⁺ and Cl⁻ forms the ionic bond that creates the crystalline structure of salt.
Properties of Ionic Compounds: Ionic compounds typically have high melting and boiling points, are brittle, and conduct electricity when dissolved in water or molten.
2. Covalent Bonds: Sharing is Caring
Covalent bonds form when atoms share one or more pairs of electrons to achieve a stable electron configuration. This sharing allows both atoms to effectively "fill" their valence shells, resulting in a stable molecule.
Types of Covalent Bonds:
- Single Covalent Bond: One pair of electrons is shared between two atoms. For example, in a hydrogen molecule (H₂), each hydrogen atom shares one electron with the other, creating a single covalent bond.
- Double Covalent Bond: Two pairs of electrons are shared between two atoms. For example, in an oxygen molecule (O₂), each oxygen atom shares two electrons with the other, forming a double covalent bond.
- Triple Covalent Bond: Three pairs of electrons are shared between two atoms. For example, in a nitrogen molecule (N₂), each nitrogen atom shares three electrons with the other, resulting in a very strong triple covalent bond.
Polar vs. Nonpolar Covalent Bonds: The electronegativity of the atoms involved determines the polarity of a covalent bond. Electronegativity is a measure of an atom's ability to attract electrons towards itself. If the electronegativity difference between two atoms is small (less than 0.4), the bond is considered nonpolar (equal sharing). If the difference is significant (greater than 0.4), the bond is polar (unequal sharing), resulting in a slightly positive end and a slightly negative end.
Examples: Water (H₂O) has two polar covalent bonds between oxygen and hydrogen, while methane (CH₄) has four nonpolar covalent bonds between carbon and hydrogen.
Properties of Covalent Compounds: Covalent compounds typically have lower melting and boiling points than ionic compounds. They may be solids, liquids, or gases at room temperature, and generally do not conduct electricity.
3. Metallic Bonds: A Sea of Electrons
Metallic bonds occur in metals, where valence electrons are delocalized and shared among a lattice of metal atoms. This creates a "sea" of mobile electrons that can move freely throughout the metal structure. The strong electrostatic attraction between the positively charged metal ions and the negatively charged electron sea holds the metal together.
Properties of Metallic Compounds: Metals are typically good conductors of heat and electricity due to the mobility of their valence electrons. They are often malleable (can be hammered into sheets) and ductile (can be drawn into wires). This is because the delocalized electrons allow metal atoms to slide past each other without disrupting the metallic bond.
Beyond the Basics: Factors Influencing Bond Formation
While the octet rule and the three main bond types offer a solid foundation, several other factors influence bond formation:
- Electronegativity: As previously mentioned, the difference in electronegativity between atoms significantly impacts the nature of the bond. A large electronegativity difference leads to ionic bonds, while a small difference leads to covalent bonds.
- Atomic Size: The size of atoms affects the distance between nuclei and influences the strength of the bond. Smaller atoms tend to form stronger bonds.
- Bond Energy: This is the amount of energy required to break a bond. Stronger bonds have higher bond energies.
- Bond Length: This is the distance between the nuclei of two bonded atoms. Shorter bond lengths generally indicate stronger bonds.
- Resonance: In some molecules, electrons can be delocalized over multiple atoms, creating resonance structures. This delocalization stabilizes the molecule and influences its properties.
- Hydrogen Bonding: A special type of dipole-dipole interaction, hydrogen bonds are relatively strong intermolecular forces that occur between molecules containing hydrogen atoms bonded to highly electronegative atoms like oxygen, nitrogen, or fluorine. These bonds play crucial roles in biological systems.
- Van der Waals Forces: These are weak intermolecular forces that arise from temporary fluctuations in electron distribution around atoms and molecules. While individually weak, they can have a cumulative effect, especially in large molecules.
The Importance of Chemical Bonding in the World Around Us
Chemical bonding is not just an abstract concept; it’s the foundation of the material world. Understanding chemical bonds allows us to comprehend:
- The properties of materials: The type of bonding directly influences the physical and chemical properties of a substance – its hardness, melting point, conductivity, reactivity, and more.
- Chemical reactions: Chemical reactions involve the breaking and formation of chemical bonds. Understanding bonding helps us predict the outcome of reactions and design new materials.
- Biological systems: Life itself depends on the intricate network of chemical bonds that hold biological molecules together. From DNA to proteins, understanding bonding is critical to understanding biology.
- Technological advancements: Many technological advancements rely on our ability to manipulate chemical bonds, creating new materials with desired properties (e.g., polymers, semiconductors, etc.).
Conclusion: A Continuous Quest for Stability
The driving force behind atom bonding is the pursuit of stability. Atoms strive to achieve a more stable electron configuration, often resembling that of a noble gas. This fundamental principle leads to diverse types of bonds – ionic, covalent, and metallic – each with its unique characteristics and consequences for the macroscopic properties of matter. The world we inhabit is a testament to the power and elegance of chemical bonding, shaping everything from the smallest molecules to the largest structures in the universe. Further research continually unravels the complexities and subtleties of these interactions, deepening our understanding of the natural world and paving the way for technological innovation.
Latest Posts
Latest Posts
-
What Is A Wet Mount Slide
Apr 03, 2025
-
Examples Of Stoichiometry In Real Life
Apr 03, 2025
-
Why Is Water Necessary For Biological Organisms
Apr 03, 2025
-
How Is Probability Used In Genetics
Apr 03, 2025
-
Liquid To Gas Endothermic Or Exothermic
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Why Do Atoms Form Bonds With Other Atoms . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
