Do Ionic Compounds Have High Boiling Points
Muz Play
Apr 05, 2025 · 5 min read
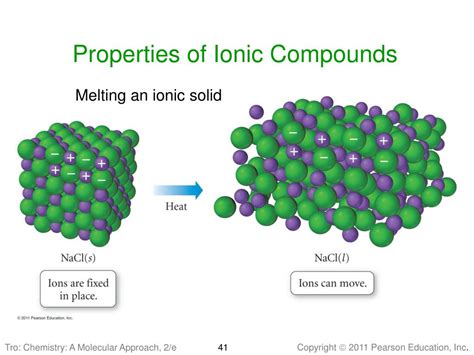
Table of Contents
Do Ionic Compounds Have High Boiling Points? A Deep Dive into Ionic Bonding and its Properties
Ionic compounds are renowned for their high boiling points. But why? This isn't just a matter of memorizing a fact; understanding the underlying reasons requires delving into the nature of ionic bonding and the forces that hold these compounds together. This comprehensive guide will explore the relationship between ionic bonding, intermolecular forces, and the resulting high boiling points observed in ionic compounds. We'll also examine exceptions and compare ionic compounds to other types of compounds.
Understanding Ionic Bonding: The Foundation of High Boiling Points
At the heart of this phenomenon lies the electrostatic attraction between oppositely charged ions. Ionic compounds are formed when atoms of elements with significantly different electronegativities react. Highly electronegative atoms, typically nonmetals, readily gain electrons to achieve a stable electron configuration, forming negatively charged ions called anions. Conversely, less electronegative atoms, often metals, readily lose electrons, forming positively charged ions called cations.
This transfer of electrons creates a strong electrostatic force of attraction between the cations and anions, forming an ionic bond. This isn't a sharing of electrons like in covalent bonds; it's a complete transfer, resulting in a strong electrostatic interaction. The strength of this interaction is directly proportional to the charges of the ions and inversely proportional to the distance between them (Coulomb's Law).
Coulomb's Law and its Influence
Coulomb's Law is crucial in understanding the strength of ionic bonds. The law states that the force of attraction (or repulsion) between two charged particles is directly proportional to the product of their charges and inversely proportional to the square of the distance between them. This means:
- Higher charges lead to stronger bonds: Compounds with ions carrying higher charges (e.g., +2 and -2) will have stronger ionic bonds than those with +1 and -1 charges.
- Smaller ions lead to stronger bonds: Smaller ions are closer together, reducing the distance between the charges and therefore increasing the strength of the electrostatic attraction.
This explains why compounds like magnesium oxide (MgO), with Mg²⁺ and O²⁻ ions, have significantly higher boiling points than sodium chloride (NaCl), with Na⁺ and Cl⁻ ions. The higher charges in MgO result in stronger electrostatic forces.
The Role of Lattice Energy in Boiling Point Elevation
The strong electrostatic forces in ionic compounds aren't just localized between individual ion pairs; they extend throughout the entire crystal lattice. A crystal lattice is a three-dimensional arrangement of ions, with each cation surrounded by several anions and vice versa. The energy required to completely separate one mole of a solid ionic compound into its gaseous ions is called lattice energy.
High lattice energy is a direct consequence of the strong electrostatic forces within the ionic lattice. Breaking these strong bonds requires a substantial amount of energy, translating directly into a high boiling point. The stronger the ionic bond (higher charge, smaller ions), the higher the lattice energy and consequently, the higher the boiling point.
Lattice Energy and its Correlation with Boiling Point
The magnitude of lattice energy is a key determinant of an ionic compound's boiling point. The stronger the electrostatic attractions, the more energy is needed to overcome them during the phase transition from solid to liquid (melting point) and from liquid to gas (boiling point). The high lattice energy is why ionic compounds are generally solids at room temperature and require significant energy input (high temperature) to transition to the gaseous phase.
Comparing Ionic Compounds to Other Compound Types
To further appreciate the high boiling points of ionic compounds, let's compare them to other types of compounds:
Covalent Compounds:
Covalent compounds are formed by sharing electrons between atoms. The intermolecular forces in covalent compounds are much weaker than the ionic bonds in ionic compounds. These forces, such as van der Waals forces, hydrogen bonding, and dipole-dipole interactions, are significantly less effective in holding molecules together. As a result, covalent compounds generally have much lower boiling points than ionic compounds.
Metallic Compounds:
Metallic compounds have a unique type of bonding involving the delocalization of electrons among a lattice of metal atoms. While the strength of metallic bonds varies widely depending on the metal, they are generally weaker than ionic bonds. Therefore, metallic compounds typically have lower boiling points than ionic compounds, though some exceptions exist with high-melting point metals like tungsten.
Exceptions to the Rule: Factors Influencing Boiling Points
While generally true, there are exceptions to the statement that all ionic compounds have extremely high boiling points. Several factors can influence the boiling point of ionic compounds:
- Ion Size: As mentioned earlier, smaller ions lead to stronger ionic bonds and higher boiling points. Larger ions, with their increased distance between charges, will have weaker bonds and lower boiling points.
- Charge Density: The charge density, or the charge per unit volume of an ion, influences the strength of the ionic bond. Higher charge density leads to stronger bonds and higher boiling points.
- Polarizability of Ions: Large, easily polarizable anions can lead to additional interionic interactions, slightly increasing the boiling point. However, this effect is generally smaller than the impact of ion size and charge.
Practical Applications and Examples
The high boiling points of ionic compounds have numerous practical applications. For example, many salts are used as heat transfer fluids in industrial processes due to their ability to withstand high temperatures without vaporizing. The high melting and boiling points also make them suitable for applications requiring thermal stability.
Conclusion: High Boiling Points – A Hallmark of Ionic Compounds
In conclusion, the high boiling points of ionic compounds are a direct consequence of the strong electrostatic attraction between oppositely charged ions, creating a high lattice energy. This strong bonding requires significant energy input to overcome, resulting in the elevated boiling points characteristic of ionic compounds. While some exceptions exist based on ion size, charge, and polarizability, the general principle remains firmly established. Understanding this fundamental relationship between ionic bonding and boiling point is crucial in various scientific and engineering fields. The ability to predict and manipulate the properties of ionic compounds based on their bonding characteristics is vital for materials science, chemistry, and other related disciplines.
Latest Posts
Latest Posts
-
Term Used For Compounds With Properties For Neutralizing Acids
Apr 05, 2025
-
How To Find Average Acceleration From Velocity Time Graph
Apr 05, 2025
-
Newtons Second Law In Rotational Form
Apr 05, 2025
-
The Citric Acid Cycle Is A Stage Of Catabolism
Apr 05, 2025
-
Which Of The Following Can Exist As A Meso Isomer
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Do Ionic Compounds Have High Boiling Points . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
