Do Solid Ionic Compounds Conduct Electricity
Muz Play
Apr 05, 2025 · 5 min read
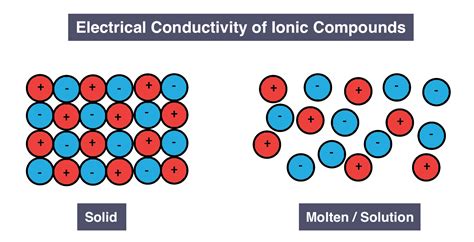
Table of Contents
Do Solid Ionic Compounds Conduct Electricity?
The question of whether solid ionic compounds conduct electricity is a fundamental concept in chemistry. The short answer is no, solid ionic compounds generally do not conduct electricity. However, understanding why this is the case requires delving into the nature of ionic compounds and the mechanism of electrical conductivity. This article will explore this topic in detail, examining the structure of ionic compounds, the role of ions and their mobility, and the conditions under which ionic compounds can conduct electricity.
The Structure of Ionic Compounds
Ionic compounds are formed through the electrostatic attraction between oppositely charged ions. These ions are created when atoms either lose or gain electrons, forming positively charged cations (like Na⁺) and negatively charged anions (like Cl⁻). The strong electrostatic forces between these ions result in a highly ordered, crystalline structure known as a lattice.
The Crystalline Lattice: A Rigid Structure
Imagine a three-dimensional arrangement of alternating cations and anions, tightly packed together. This is the essence of an ionic lattice. The ions are not free to move around; they are held firmly in place by the strong electrostatic forces. This rigid structure is responsible for many of the properties of ionic compounds, including their high melting and boiling points and their brittleness.
Strong Electrostatic Forces: Immobile Ions
The strong electrostatic forces that bind the ions together are crucial in understanding why solid ionic compounds don't conduct electricity. For electrical conductivity, charged particles—in this case, ions—need to be able to move freely. In a solid ionic lattice, this movement is severely restricted. The ions are locked in their positions, unable to migrate through the structure to carry an electric current.
Electrical Conductivity: The Movement of Charge Carriers
Electrical conductivity is the ability of a material to allow the flow of electric current. This flow is facilitated by the movement of charge carriers, which can be electrons or ions. Metals, for instance, are excellent conductors because they have delocalized electrons that can move freely throughout the metal lattice.
The Role of Mobile Charge Carriers
In ionic compounds, the potential charge carriers are the ions themselves. However, in the solid state, these ions are immobile. Therefore, even though there are abundant charges within the lattice, they cannot contribute to the flow of electricity. The ions are effectively "stuck" within their fixed positions in the crystal structure.
Contrast with Metallic Conductivity
It's important to contrast this with the mechanism of electrical conductivity in metals. In metals, the valence electrons are delocalized and form a "sea" of electrons that can move freely throughout the metal lattice. This free movement of electrons allows for the efficient flow of electric current, making metals excellent conductors.
When Ionic Compounds Do Conduct Electricity: The Role of the Molten State and Solutions
While solid ionic compounds are poor conductors, their conductivity changes dramatically when they are melted or dissolved in water.
Molten Ionic Compounds: Increased Ion Mobility
When an ionic compound is melted, the strong electrostatic forces holding the ions in place are overcome by the increased kinetic energy of the ions. This causes the lattice structure to break down, and the ions become mobile. These freely moving ions can now carry an electric current, making molten ionic compounds good conductors of electricity.
Aqueous Solutions of Ionic Compounds: Ion Dissociation
Similarly, when an ionic compound dissolves in water, it dissociates into its constituent ions. The water molecules surround the ions, reducing the strong electrostatic attractions between them and allowing them to move independently. This again leads to mobile charge carriers, resulting in an aqueous solution that conducts electricity. The higher the concentration of dissolved ions, the higher the conductivity.
Electrolytes: Conducting Solutions
Solutions that conduct electricity due to the presence of dissolved ions are called electrolytes. These solutions are crucial in many applications, including batteries, electroplating, and biological systems.
Factors Affecting Conductivity in Molten and Dissolved Ionic Compounds
Several factors influence the electrical conductivity of molten and dissolved ionic compounds:
-
Temperature: Higher temperatures generally increase conductivity in molten ionic compounds because they increase the kinetic energy of the ions, making them move faster and more freely. In solutions, temperature effects are more complex and depend on the specific solute and solvent.
-
Concentration: In solutions, higher concentrations of dissolved ions lead to higher conductivity because there are more charge carriers available to carry the current.
-
Nature of the Ions: The size and charge of the ions can affect their mobility and hence the conductivity. Smaller ions with higher charges generally have higher mobility.
-
Solvent: In solutions, the nature of the solvent plays a crucial role. Water, being a polar solvent, is particularly effective at dissolving ionic compounds and facilitating ion mobility. Nonpolar solvents, on the other hand, do not dissolve ionic compounds well and therefore result in poor conductivity.
Applications of Ionic Conductivity
The conductivity of molten and dissolved ionic compounds has many important applications:
-
Electrolysis: The process of using electricity to drive non-spontaneous chemical reactions, such as the decomposition of water into hydrogen and oxygen, relies on the conductivity of molten or dissolved ionic compounds.
-
Batteries: Batteries use the movement of ions between electrodes to generate an electric current. The electrolyte, often an aqueous or non-aqueous solution of dissolved salts, is essential for this process.
-
Electroplating: The process of coating a metal surface with another metal using an electric current relies on the conductivity of the electrolyte solution.
-
Corrosion Prevention: Controlling the conductivity of the surrounding environment can help prevent corrosion of metal surfaces.
-
Biological Systems: The conductivity of body fluids, which contain many dissolved ions, is crucial for the functioning of nerves and muscles.
Conclusion: Conductivity Depends on Ion Mobility
In summary, solid ionic compounds do not conduct electricity because their ions are immobile within the rigid crystal lattice. However, when the ionic compound is melted or dissolved in a suitable solvent, the ions become mobile, leading to electrical conductivity. This difference in conductivity highlights the crucial role of ion mobility in electrical conduction. The applications of this conductivity, ranging from industrial processes to biological functions, underline its importance in various fields of science and technology. Understanding the relationship between ionic structure, ion mobility, and electrical conductivity is fundamental to appreciating the behavior of ionic compounds and their diverse applications.
Latest Posts
Latest Posts
-
Newtons Second Law In Rotational Form
Apr 05, 2025
-
The Citric Acid Cycle Is A Stage Of Catabolism
Apr 05, 2025
-
Which Of The Following Can Exist As A Meso Isomer
Apr 05, 2025
-
In A Chemical Reaction Atoms Are
Apr 05, 2025
-
What Is The Unit Of Angular Momentum
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about Do Solid Ionic Compounds Conduct Electricity . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
