Draw A Structural Formula For 3-bromo-4-chloro-1 1-dimethylcyclohexane
Muz Play
Apr 02, 2025 · 5 min read
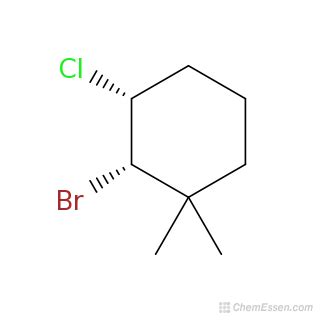
Table of Contents
Drawing the Structural Formula for 3-Bromo-4-chloro-1,1-dimethylcyclohexane: A Step-by-Step Guide
Understanding organic chemistry often hinges on visualizing molecules. This article provides a comprehensive guide to drawing the structural formula for 3-bromo-4-chloro-1,1-dimethylcyclohexane, breaking down the process step-by-step and explaining the underlying principles of nomenclature. We'll cover everything from understanding the IUPAC name to creating a clear and accurate representation of the molecule's structure.
Deconstructing the IUPAC Name: A Key to Understanding the Structure
The IUPAC (International Union of Pure and Applied Chemistry) name, 3-bromo-4-chloro-1,1-dimethylcyclohexane, gives us a blueprint for constructing the molecule. Let's dissect it piece by piece:
-
Cyclohexane: This is the parent hydrocarbon, a six-carbon ring forming a cyclic structure. Imagine a hexagon – that's your starting point.
-
1,1-dimethyl: This indicates two methyl groups (CH₃) attached to the same carbon atom on the cyclohexane ring. We'll need to identify carbon #1 to place these methyl groups correctly.
-
3-bromo: This signifies a bromine atom (Br) attached to the third carbon atom of the cyclohexane ring.
-
4-chloro: This tells us a chlorine atom (Cl) is attached to the fourth carbon atom of the cyclohexane ring.
Step-by-Step Drawing Process: From Name to Structure
Now, let's translate the name into a structural formula:
Step 1: Drawing the Cyclohexane Ring
Begin by drawing a hexagon representing the cyclohexane ring. Remember that each corner of the hexagon represents a carbon atom, and each carbon atom has two hydrogen atoms attached (unless replaced by another substituent). For simplicity, we might omit these hydrogen atoms in the initial drawing, but we'll need to remember their presence.
1
/ \
2---3
| |
6---4
\ /
5
Step 2: Locating Carbon #1 and Adding the Dimethyl Groups
We need to choose a carbon atom to be carbon #1. The position of the substituents (bromo and chloro) are relative to this carbon. It doesn't strictly matter which carbon you choose as #1, but consistency is key. For this example, let’s choose the top carbon (as indicated above). Now, add two methyl groups (CH₃) to this carbon. This is why the name includes "1,1-dimethyl". Both methyl groups are connected to the same carbon atom (carbon #1).
1
/|\
2---3
| |
6---4
\ /
5
CH3 CH3
Step 3: Adding the Bromo and Chloro Substituents
According to the name, we have a bromine atom at carbon #3 and a chlorine atom at carbon #4. Locate these carbons on your ring and add the respective atoms:
1
/|\
2---3-Br
| |
6---4-Cl
\ /
5
CH3 CH3
Step 4: Adding Implicit Hydrogens (Optional but Recommended)
While often omitted for brevity, it is beneficial, especially for beginners, to explicitly include the hydrogen atoms attached to each carbon atom. Remember each carbon needs four bonds. Count the number of bonds to each carbon. If the number is less than four, add hydrogen atoms to complete the tetravalency of the carbon.
1
/|\
2---3-Br
| |
6---4-Cl
\ /
5
CH3 CH3
H H H H H H
The final structure should show each carbon atom with four bonds. This comprehensive representation ensures the accuracy of the molecular structure and understanding.
Advanced Considerations: Conformations and Stereoisomers
Cyclohexane rings don't exist as flat hexagons; they adopt various conformations to minimize steric hindrance (repulsion between atoms). The most stable conformations are the chair and boat conformations. The molecule 3-bromo-4-chloro-1,1-dimethylcyclohexane can exist in multiple chair conformations, each with slightly different energies.
Furthermore, the presence of chiral centers (carbon atoms bonded to four different groups) can lead to stereoisomers. While this molecule doesn't inherently contain a chiral center, the presence of multiple substituents emphasizes the importance of considering spatial arrangement in complex organic molecules. Drawing the molecule in a 3D representation helps visualize the molecule's spatial structure more accurately.
Importance of Accurate Structural Formula Representation
Accurate representation of the structural formula is crucial in organic chemistry for several reasons:
-
Understanding Reactivity: The spatial arrangement of atoms significantly influences a molecule's reactivity. Knowing the exact structure helps predict how a molecule will behave in chemical reactions.
-
Naming and Classification: The IUPAC naming system is directly tied to the molecule's structure. An accurate structural formula allows for unambiguous naming.
-
Physical Properties: The structural formula helps understand the molecule's physical properties, such as melting point, boiling point, and solubility.
Applications and Further Exploration
Understanding the structural formula of 3-bromo-4-chloro-1,1-dimethylcyclohexane serves as a foundational skill for more advanced concepts in organic chemistry. It forms the basis for understanding reactions involving substitution, elimination, and other transformations common in organic synthesis.
Further exploration could include:
-
Spectral Analysis: Analyzing the NMR (Nuclear Magnetic Resonance) and IR (Infrared) spectra of this compound to correlate the predicted structure with experimental data.
-
Reaction Mechanisms: Exploring how this molecule might react with different reagents under various conditions.
-
Computational Chemistry: Using computational tools to calculate molecular properties and optimize the geometry of the molecule.
By following these steps and understanding the underlying principles, you can accurately draw the structural formula for 3-bromo-4-chloro-1,1-dimethylcyclohexane and confidently approach the structural representation of other organic molecules. Remember, practice makes perfect! The more you practice drawing these structures, the more confident and efficient you will become in understanding organic chemistry. The ability to visualize and interpret molecular structures is a fundamental skill for success in this fascinating field.
Latest Posts
Latest Posts
-
Which Of The Following Is A Single Replacement Reaction
Apr 03, 2025
-
Point Estimate Of The Population Standard Deviation
Apr 03, 2025
-
A Magnifier Makes Things Appear Larger Because
Apr 03, 2025
-
What Is The Difference Between Intermolecular And Intramolecular Forces
Apr 03, 2025
-
Where Is The Energy Stored In Glucose
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Draw A Structural Formula For 3-bromo-4-chloro-1 1-dimethylcyclohexane . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
