Draw The Tautomer Of This Aldehyde
Muz Play
Apr 02, 2025 · 6 min read
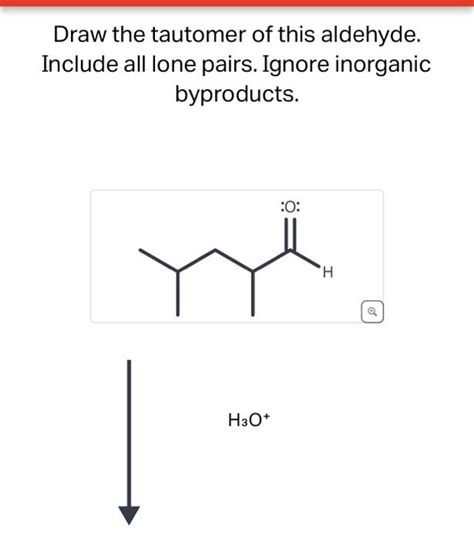
Table of Contents
Drawing the Tautomers of Aldehydes: A Comprehensive Guide
Aldehydes, a fundamental class of organic compounds, possess a unique characteristic: tautomerism. Understanding tautomerism is crucial for predicting chemical reactivity and interpreting spectroscopic data. This article delves deep into the tautomerism of aldehydes, focusing on how to draw and understand the different tautomeric forms, exploring the underlying principles, and providing practical examples to solidify your knowledge.
What is Tautomerism?
Tautomerism refers to the rapid interconversion of two isomers—tautomers—that differ in the position of a proton and a double bond. This dynamic equilibrium is typically catalyzed by either acid or base. The most common type of tautomerism is keto-enol tautomerism, and aldehydes are capable of exhibiting this phenomenon.
Keto-Enol Tautomerism in Aldehydes
Aldehydes, containing a carbonyl group (C=O) directly bonded to a hydrogen atom, can exist in two tautomeric forms: the keto form and the enol form.
-
Keto Form: This is the more stable and predominantly observed form of an aldehyde. It features a carbonyl group (C=O) and a carbon-hydrogen (C-H) bond.
-
Enol Form: The enol form contains a hydroxyl group (-OH) attached to a carbon atom double-bonded to another carbon atom (C=C). This configuration results in a carbon-carbon double bond and an alcohol functional group.
The interconversion between the keto and enol forms involves the movement of a proton (H⁺) and the repositioning of a double bond. This process is catalyzed by either acidic or basic conditions.
Drawing the Enol Tautomer
Let's break down the process of drawing the enol tautomer from a given aldehyde:
-
Identify the alpha-carbon: This is the carbon atom directly adjacent to the carbonyl carbon.
-
Move a proton: Transfer the hydrogen atom from the alpha-carbon to the oxygen atom of the carbonyl group.
-
Form a double bond: Create a double bond between the alpha-carbon and the carbonyl carbon.
Example 1: Formaldehyde (HCHO)
The simplest aldehyde, formaldehyde (HCHO), can also undergo tautomerism, although the enol form is highly unstable in this case.
-
Keto form: HCHO (a single carbon with a double bond to oxygen and a single bond to hydrogen)
-
Enol form: It is highly unstable but can be conceptually drawn as H₂C=C(OH)₂. Note that both hydrogens are connected to the same carbon. This highly unstable di-hydroxymethylene is an exception and generally does not participate in typical enol-keto equilibrium.
Example 2: Acetaldehyde (CH₃CHO)
Acetaldehyde (CH₃CHO) provides a clearer example.
-
Keto form: CH₃CHO (methyl group linked to a carbonyl and a hydrogen)
-
Enol form: CH₂=CHOH (a double bond between two carbons, with a hydroxyl group (-OH) on one of them). Note the shift of a proton (H⁺) from the alpha-carbon to the oxygen atom, and the formation of the C=C double bond.
Example 3: Propanal (CH₃CH₂CHO)
Propanal allows a more visual demonstration of the process.
-
Keto form: CH₃CH₂CHO (an ethyl group attached to a carbonyl and a hydrogen)
-
Enol form: CH₃CH=CHOH (a methyl group, a double bond between the second and third carbon, and a hydroxyl group on the third carbon). Again, note the proton shift and double bond formation.
Example 4: More complex aldehydes
The principle remains consistent even with more complex aldehydes containing substituents. The proton transfer always occurs from the alpha-carbon, and the double bond forms between the alpha-carbon and the carbonyl carbon.
Consider an aldehyde with a substituent on the alpha-carbon: R-CH(R')-CHO, where R and R' are alkyl groups. The enol form would be R-C(R')=CHOH.
Factors Affecting Keto-Enol Equilibrium
The relative stability of the keto and enol forms depends on several factors:
-
Steric effects: Bulky substituents near the double bond in the enol form can destabilize it due to steric hindrance.
-
Electronic effects: Electron-withdrawing groups on the alpha-carbon can stabilize the enol form by delocalizing the negative charge on the oxygen.
-
Hydrogen bonding: Intramolecular hydrogen bonding in the enol form can increase its stability.
-
Solvent effects: Polar solvents tend to favor the more polar enol form, while nonpolar solvents generally favor the keto form.
Generally, the keto form is significantly more stable than the enol form in simple aldehydes. However, the equilibrium constant (K) varies substantially depending on the specific structure and conditions.
Acid and Base Catalysis of Tautomerism
The interconversion between keto and enol forms is not spontaneous; it requires catalysis.
Acid Catalysis
In acid catalysis, a proton from the acid adds to the carbonyl oxygen, increasing the electrophilicity of the carbonyl carbon. This allows for a proton abstraction from the alpha-carbon, followed by a rearrangement to form the enol. The reverse reaction involves the addition of a proton to the double bond, regenerating the keto form.
Base Catalysis
In base catalysis, a base abstracts a proton from the alpha-carbon, forming an enolate ion. This enolate ion can then abstract a proton from the solvent or another molecule to generate the enol form. The reverse reaction involves protonation of the enolate ion, reforming the keto form.
Applications and Significance
Understanding aldehyde tautomerism is crucial in various fields:
-
Organic synthesis: Many organic reactions involve enol intermediates, highlighting the importance of considering tautomerism.
-
Spectroscopy: NMR and IR spectroscopy can be used to identify and quantify the keto and enol forms in equilibrium.
-
Biochemistry: Keto-enol tautomerism plays a role in enzyme mechanisms and metabolic pathways. For example, the enol form of pyruvate is crucial in several metabolic processes.
-
Medicinal chemistry: Drug design often involves manipulating the tautomeric equilibrium to optimize drug activity and bioavailability.
Advanced Concepts
While the simple keto-enol tautomerism is the most commonly encountered, some aldehydes can exhibit more complex tautomerism. For example, certain cyclic aldehydes might undergo ring-chain tautomerism, where the equilibrium exists between a cyclic form and an open-chain form. These more complex cases require a deeper understanding of organic chemistry principles.
Conclusion
Drawing the tautomer of an aldehyde involves a simple yet crucial understanding of proton transfer and double bond rearrangement. While the keto form usually predominates, the enol form plays a significant role in the reactivity and properties of aldehydes. Mastering this concept is essential for any student or professional involved in organic chemistry, biochemistry, or related fields. By understanding the underlying principles and practicing the drawing process, you can confidently navigate the world of aldehyde tautomerism. Remember to consider the various factors influencing the equilibrium and the catalytic mechanisms that drive the interconversion between keto and enol forms. This comprehensive understanding will undoubtedly enhance your problem-solving capabilities and enrich your appreciation for the dynamic nature of organic molecules.
Latest Posts
Latest Posts
-
Which State Of Matter Has A Definite Shape
Apr 03, 2025
-
Which Of The Following Is A Single Replacement Reaction
Apr 03, 2025
-
Point Estimate Of The Population Standard Deviation
Apr 03, 2025
-
A Magnifier Makes Things Appear Larger Because
Apr 03, 2025
-
What Is The Difference Between Intermolecular And Intramolecular Forces
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Draw The Tautomer Of This Aldehyde . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
