Lewis Base Vs Bronsted Lowry Base
Muz Play
Apr 02, 2025 · 6 min read
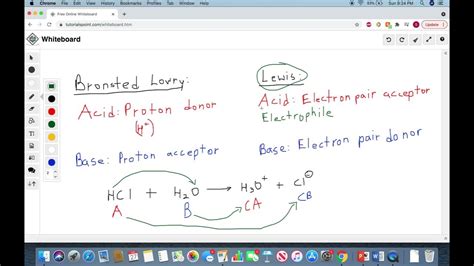
Table of Contents
Lewis Base vs. Brønsted-Lowry Base: Understanding the Differences
The concepts of acids and bases are fundamental to chemistry, underpinning numerous reactions and processes. While often used interchangeably in simpler contexts, a deeper understanding reveals subtle yet crucial distinctions between different acid-base theories. Two prominent theories, the Brønsted-Lowry and Lewis theories, offer distinct perspectives on what constitutes an acid or a base. This article delves into the key differences between Lewis bases and Brønsted-Lowry bases, highlighting their similarities, contrasting their definitions, and exploring their applications.
Defining Brønsted-Lowry Bases
The Brønsted-Lowry theory, proposed independently by Johannes Nicolaus Brønsted and Thomas Martin Lowry in 1923, defines a base as a proton acceptor. This definition centers around the transfer of a proton (H⁺ ion). A Brønsted-Lowry base reacts with an acid by accepting a proton, resulting in the formation of its conjugate acid.
Key characteristics of a Brønsted-Lowry base:
- Proton Acceptor: The defining feature is its ability to accept a proton (H⁺).
- Presence of a Lone Pair: Often, but not always, Brønsted-Lowry bases possess a lone pair of electrons to bond with the proton. This lone pair facilitates the acceptance of the proton.
- Conjugate Acid Formation: Upon accepting a proton, the base forms its conjugate acid. The conjugate acid is the species that remains after the base has accepted a proton.
Examples of Brønsted-Lowry Bases:
- Ammonia (NH₃): Ammonia readily accepts a proton to form the ammonium ion (NH₄⁺).
- Hydroxide ion (OH⁻): The hydroxide ion is a strong Brønsted-Lowry base, readily accepting a proton to form water (H₂O).
- Water (H₂O): Water can act as both an acid and a base (amphoteric), accepting a proton to form the hydronium ion (H₃O⁺).
- Carbonate ion (CO₃²⁻): Carbonate ions are strong bases, accepting protons to form bicarbonate (HCO₃⁻) and carbonic acid (H₂CO₃).
Defining Lewis Bases
The Lewis theory, introduced by Gilbert N. Lewis in 1923, provides a broader definition of acids and bases. A Lewis base is defined as an electron pair donor. This definition focuses on the donation of an electron pair to form a coordinate covalent bond, where both electrons in the bond come from the same atom. Unlike the Brønsted-Lowry theory, the Lewis theory does not explicitly require the transfer of a proton.
Key characteristics of a Lewis base:
- Electron Pair Donor: The crucial feature is the ability to donate a pair of electrons.
- Formation of a Coordinate Covalent Bond: The Lewis base donates its electron pair to form a coordinate covalent bond with a Lewis acid.
- No Proton Requirement: Proton transfer is not a necessary condition for a substance to be classified as a Lewis base.
Examples of Lewis Bases:
- Ammonia (NH₃): Ammonia possesses a lone pair of electrons and acts as a Lewis base by donating this pair to form a coordinate covalent bond.
- Water (H₂O): Similar to ammonia, water's lone pairs allow it to act as a Lewis base.
- Chloride ion (Cl⁻): The chloride ion, with its lone pairs, can donate electrons to act as a Lewis base.
- Carbon monoxide (CO): Carbon monoxide, with its lone pairs on the oxygen atom, can act as a Lewis base. This is important in its coordination with transition metals.
Key Differences between Lewis and Brønsted-Lowry Bases
The most significant difference lies in their definitions:
| Feature | Brønsted-Lowry Base | Lewis Base |
|---|---|---|
| Definition | Proton (H⁺) acceptor | Electron pair donor |
| Proton Transfer | Essential | Not essential |
| Scope | Narrower | Broader |
| Bonding | Usually involves covalent bond formation | Always involves coordinate covalent bond |
While all Brønsted-Lowry bases are also Lewis bases (because proton acceptance necessitates electron pair donation), the reverse is not true. Many Lewis bases do not involve proton transfer and therefore don't fit the Brønsted-Lowry definition.
Examples Illustrating the Differences
Let's consider a few examples to highlight the differences:
1. Ammonia (NH₃) reacting with HCl:
- Brønsted-Lowry: NH₃ accepts a proton from HCl, forming NH₄⁺ (conjugate acid) and Cl⁻.
- Lewis: NH₃ donates its lone pair of electrons to the H⁺ ion from HCl, forming a coordinate covalent bond in NH₄⁺. Here, both definitions apply.
2. Trimethylamine (N(CH₃)₃) reacting with BF₃:
- Brønsted-Lowry: This reaction doesn't involve proton transfer. Therefore, it cannot be explained using the Brønsted-Lowry theory.
- Lewis: N(CH₃)₃ donates its lone pair of electrons to the boron atom in BF₃, forming a coordinate covalent bond. This reaction is easily explained by the Lewis theory. BF₃ acts as a Lewis acid (electron pair acceptor).
3. The reaction between AlCl₃ and Cl⁻:
- Brønsted-Lowry: No proton transfer occurs.
- Lewis: Cl⁻ donates its electron pair to the electron-deficient aluminum atom in AlCl₃, forming AlCl₄⁻. AlCl₃ acts as a Lewis acid.
Applications and Significance
The expanded scope of the Lewis theory is crucial for understanding a wide range of reactions in organic and inorganic chemistry, particularly those involving transition metal complexes. Many reactions in coordination chemistry rely on the Lewis acid-base interactions.
Applications of Lewis Acid-Base Theory:
- Catalysis: Many catalysts function through Lewis acid-base interactions. Lewis acids can activate reactants by coordinating with them, making them more susceptible to attack by other reagents.
- Coordination Chemistry: The formation of coordination complexes is fundamentally based on Lewis acid-base interactions. Transition metal ions, acting as Lewis acids, coordinate with ligands (Lewis bases) to form stable complexes.
- Organic Chemistry: Numerous organic reactions, such as the Friedel-Crafts alkylation and acylation, involve Lewis acid catalysts that activate electrophilic reagents.
- Biochemistry: Many biological processes involve Lewis acid-base interactions. For example, enzyme-substrate interactions often involve coordination bonds.
Applications of Brønsted-Lowry Theory:
- Acid-Base Titrations: The Brønsted-Lowry theory forms the basis for acid-base titrations, a crucial analytical technique.
- Buffer Solutions: Understanding conjugate acid-base pairs is crucial for preparing and utilizing buffer solutions to maintain a stable pH.
- General Chemistry: The Brønsted-Lowry theory is often the introductory acid-base theory taught in general chemistry, providing a simpler framework for understanding many common reactions.
Conclusion
While both Brønsted-Lowry and Lewis theories describe acids and bases, the Lewis theory offers a broader and more inclusive perspective. The Brønsted-Lowry theory focuses specifically on proton transfer, limiting its applicability to reactions involving protons. In contrast, the Lewis theory encompasses a wider range of reactions, including those without proton transfer, making it essential for understanding many chemical processes, particularly in coordination chemistry and catalysis. Both theories are valuable tools for understanding the behavior of acids and bases, with the choice of which theory to apply depending on the specific chemical system under consideration. Understanding the nuances and differences between these theories is crucial for a comprehensive grasp of acid-base chemistry.
Latest Posts
Latest Posts
-
Which Of The Following Is A Single Replacement Reaction
Apr 03, 2025
-
Point Estimate Of The Population Standard Deviation
Apr 03, 2025
-
A Magnifier Makes Things Appear Larger Because
Apr 03, 2025
-
What Is The Difference Between Intermolecular And Intramolecular Forces
Apr 03, 2025
-
Where Is The Energy Stored In Glucose
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Lewis Base Vs Bronsted Lowry Base . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
