Reacts With Acid Physical Or Chemical
Muz Play
Apr 02, 2025 · 6 min read
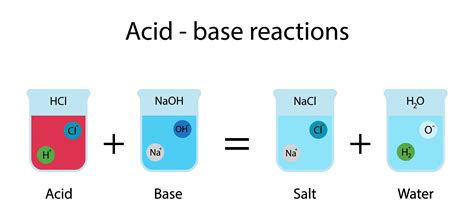
Table of Contents
Reactions with Acid: A Deep Dive into Physical and Chemical Changes
Acids, with their characteristic sour taste and ability to react with bases, play a crucial role in countless chemical reactions. Understanding how acids interact with different substances, particularly whether the interaction is physical or chemical, is fundamental to chemistry and numerous applications, from industrial processes to biological systems. This comprehensive article will explore the nuances of reactions with acids, differentiating between physical and chemical changes, providing illustrative examples, and examining the underlying principles.
Defining Acids and Their Properties
Before delving into reactions, let's establish a clear understanding of acids. Acids are substances that donate protons (H⁺ ions) in a chemical reaction, according to the Brønsted-Lowry definition. This proton donation is the key characteristic that governs their reactivity. The strength of an acid depends on its tendency to donate protons; strong acids readily donate protons, while weak acids only partially dissociate. Common examples of acids include hydrochloric acid (HCl), sulfuric acid (H₂SO₄), nitric acid (HNO₃), and acetic acid (CH₃COOH).
Acids exhibit several characteristic properties:
- Sour taste: A distinctive property, although it shouldn't be tested directly due to potential dangers.
- Reaction with bases: Acids neutralize bases in a characteristic acid-base reaction, forming salt and water.
- Reaction with metals: Many acids react with metals, producing hydrogen gas and a salt.
- Change in pH: Acids lower the pH of a solution, indicating increased acidity.
- Electrolytic properties: Acids conduct electricity when dissolved in water due to the presence of ions.
Physical Changes vs. Chemical Changes
Understanding the difference between physical and chemical changes is paramount when analyzing reactions with acids.
Physical changes alter the physical properties of a substance, like its shape, size, or state of matter, without changing its chemical composition. These changes are often reversible. Examples include melting ice, dissolving sugar in water, or crushing a rock.
Chemical changes, also known as chemical reactions, involve the rearrangement of atoms and molecules to form new substances with different chemical properties. These changes are usually irreversible and often accompanied by observable phenomena like color change, gas evolution, or precipitation.
Reactions with Acids: Physical Changes
While many acid reactions are chemical in nature, some interactions result in physical changes. A key example is the dissolution of a non-reactive substance in an acid.
Dissolution of Non-Reactive Substances
Certain substances, like sugar or many salts, dissolve in acids without undergoing a chemical change. The acid acts as a solvent, separating the molecules or ions of the solute, but the chemical composition of both the solute and the acid remains unchanged. This is a physical change because the process is reversible (the solute can be recovered through evaporation).
For example, dissolving table salt (NaCl) in hydrochloric acid doesn't chemically alter the salt or the acid. The NaCl dissociates into Na⁺ and Cl⁻ ions, surrounded by the acid molecules, but no new chemical bonds are formed. Evaporation of the acid would recover the solid NaCl.
Reactions with Acids: Chemical Changes
The majority of reactions involving acids are chemical in nature, leading to the formation of new substances. These chemical reactions can be categorized into various types:
1. Acid-Base Neutralization Reactions
This is perhaps the most characteristic reaction of acids. When an acid reacts with a base, they neutralize each other, producing salt and water. The reaction is exothermic, releasing heat.
Example: The reaction between hydrochloric acid (HCl) and sodium hydroxide (NaOH) produces sodium chloride (NaCl) and water (H₂O):
HCl(aq) + NaOH(aq) → NaCl(aq) + H₂O(l)
This reaction is a classic example of a double displacement reaction, where the ions exchange partners.
2. Reactions with Metals
Many acids, especially strong acids, react with active metals (metals located higher in the reactivity series than hydrogen) to produce hydrogen gas and a salt. This reaction is also exothermic.
Example: The reaction between hydrochloric acid (HCl) and zinc (Zn) produces zinc chloride (ZnCl₂) and hydrogen gas (H₂):
2HCl(aq) + Zn(s) → ZnCl₂(aq) + H₂(g)
This is a single displacement reaction, where the hydrogen in the acid is replaced by the zinc. The reactivity of the metal determines the vigor of the reaction. Less reactive metals, such as copper, do not readily react with many acids.
3. Reactions with Carbonates and Bicarbonates
Acids react with carbonates and bicarbonates to produce carbon dioxide gas, water, and a salt. The effervescence (fizzing) caused by the release of CO₂ is a characteristic observation.
Example: The reaction between hydrochloric acid (HCl) and calcium carbonate (CaCO₃) produces calcium chloride (CaCl₂), carbon dioxide (CO₂), and water (H₂O):
2HCl(aq) + CaCO₃(s) → CaCl₂(aq) + CO₂(g) + H₂O(l)
This is another example of a double displacement reaction, followed by the decomposition of carbonic acid (H₂CO₃) into CO₂ and H₂O.
4. Reactions with Metal Oxides
Metal oxides, which are basic in nature, react with acids to form salt and water. This is similar to an acid-base neutralization reaction.
Example: The reaction between sulfuric acid (H₂SO₄) and copper(II) oxide (CuO) produces copper(II) sulfate (CuSO₄) and water (H₂O):
H₂SO₄(aq) + CuO(s) → CuSO₄(aq) + H₂O(l)
5. Esterification Reactions (Organic Acids)
Carboxylic acids, a type of organic acid, react with alcohols in the presence of an acid catalyst to form esters and water. This reaction is an example of a condensation reaction, where water is eliminated.
Example: The reaction between acetic acid (CH₃COOH) and ethanol (CH₃CH₂OH) in the presence of sulfuric acid produces ethyl acetate (CH₃COOCH₂CH₃) and water (H₂O):
CH₃COOH(l) + CH₃CH₂OH(l) ⇌ CH₃COOCH₂CH₃(l) + H₂O(l)
This reaction is reversible and requires careful control of reaction conditions to maximize ester yield.
Identifying Physical and Chemical Changes in Acid Reactions
Distinguishing between physical and chemical changes in acid reactions requires careful observation and analysis. Key indicators of a chemical change include:
- Gas evolution: The production of bubbles indicates a gas is being formed.
- Precipitate formation: The appearance of a solid from a solution.
- Color change: A significant shift in color suggests a chemical reaction.
- Temperature change: Exothermic reactions release heat, while endothermic reactions absorb heat.
- Irreversibility: Chemical changes are typically difficult or impossible to reverse easily.
Applications of Acid Reactions
Acid reactions are fundamental to many industrial processes and have crucial roles in various fields:
- Industrial processes: Acid reactions are used in the production of fertilizers, plastics, dyes, and pharmaceuticals.
- Metallurgy: Acids are used for cleaning and etching metals.
- Food and beverage industry: Acids are used as preservatives, flavor enhancers, and leavening agents.
- Biological systems: Acids play vital roles in digestion, maintaining pH balance, and numerous metabolic processes.
Safety Precautions When Handling Acids
Acids can be corrosive and hazardous. It's crucial to always follow safety guidelines when working with acids:
- Wear appropriate safety equipment: This includes gloves, eye protection, and lab coats.
- Work in a well-ventilated area: Many acid reactions produce harmful fumes.
- Handle acids carefully: Avoid spills and contact with skin.
- Neutralize spills immediately: Use appropriate neutralizing agents.
- Dispose of acids properly: Follow established protocols for waste disposal.
Conclusion
Understanding the difference between physical and chemical changes in reactions with acids is essential for anyone working with chemicals. From acid-base neutralizations to reactions with metals and organic compounds, the diverse reactivity of acids makes them central to a wide range of applications. By carefully observing the reaction and considering the properties of the reactants and products, one can accurately identify whether the interaction is a physical or a chemical change. Always remember to prioritize safety when working with acids due to their potential hazards. This detailed exploration provides a strong foundation for further investigations into the fascinating world of acid chemistry.
Latest Posts
Latest Posts
-
Which State Of Matter Has A Definite Shape
Apr 03, 2025
-
Which Of The Following Is A Single Replacement Reaction
Apr 03, 2025
-
Point Estimate Of The Population Standard Deviation
Apr 03, 2025
-
A Magnifier Makes Things Appear Larger Because
Apr 03, 2025
-
What Is The Difference Between Intermolecular And Intramolecular Forces
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Reacts With Acid Physical Or Chemical . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
