Sodium Chloride Is An Example Of
Muz Play
Apr 02, 2025 · 7 min read
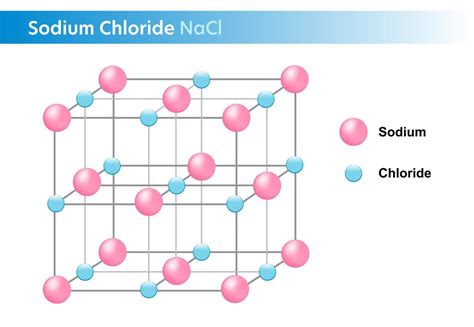
Table of Contents
Sodium Chloride: A Prime Example of Ionic Bonding, Crystal Structure, and Everyday Importance
Sodium chloride (NaCl), commonly known as table salt, is much more than just a kitchen staple. It serves as a quintessential example across several scientific disciplines, showcasing fundamental principles of chemistry, physics, and even geology. This article delves deep into the multifaceted nature of NaCl, exploring its chemical bonding, crystal structure, diverse applications, and significant role in biological systems.
Sodium Chloride as a Paragon of Ionic Bonding
At its core, sodium chloride's significance lies in its exemplary demonstration of ionic bonding. This type of chemical bond arises from the electrostatic attraction between oppositely charged ions. Sodium (Na), an alkali metal, readily loses one electron to achieve a stable electron configuration, forming a positively charged sodium ion (Na⁺). Chlorine (Cl), a halogen, readily gains one electron to achieve a stable configuration, forming a negatively charged chloride ion (Cl⁻). The strong electrostatic force of attraction between these oppositely charged ions constitutes the ionic bond that holds the sodium chloride crystal lattice together.
This electron transfer process, crucial to understanding ionic bonding, is driven by the difference in electronegativity between sodium and chlorine. Electronegativity measures an atom's ability to attract electrons within a chemical bond. Chlorine, being significantly more electronegative than sodium, exerts a stronger pull on the shared electrons, effectively "stealing" an electron from sodium. This fundamental principle governs the formation of countless ionic compounds, making NaCl a pivotal example for learning about chemical bonding.
The Role of Electrostatic Forces in NaCl Structure
The intense electrostatic attraction between Na⁺ and Cl⁻ ions isn't just about bond formation; it dictates the entire crystal structure of sodium chloride. These ions arrange themselves in a highly ordered, three-dimensional lattice structure known as a face-centered cubic (FCC) structure. This arrangement maximizes the electrostatic attraction between oppositely charged ions while minimizing repulsion between like-charged ions. Each sodium ion is surrounded by six chloride ions, and vice-versa, creating a remarkably stable and efficient packing arrangement. Understanding this structure is crucial to understanding many of NaCl's physical properties, such as its hardness and melting point.
Contrast with Covalent Bonding
It's important to contrast ionic bonding in NaCl with covalent bonding, another fundamental type of chemical bond. In covalent bonding, atoms share electrons to achieve a stable electron configuration, as seen in molecules like water (H₂O) or methane (CH₄). In contrast, ionic bonding involves a complete transfer of electrons, leading to the formation of distinct ions held together by electrostatic forces. This difference significantly impacts the properties of the resulting compounds. Ionic compounds tend to have higher melting and boiling points, are often brittle, and conduct electricity when molten or dissolved in water, unlike most covalent compounds.
The Crystalline Structure of Sodium Chloride: A Detailed Look
The face-centered cubic structure of sodium chloride is not just a random arrangement; it's a precise and repeating pattern that extends throughout the entire crystal. This highly ordered structure is responsible for many of NaCl's macroscopic properties. The regularity of the lattice leads to its characteristic cleavage—the tendency to break along specific planes, yielding flat surfaces. This is a direct consequence of the strong ionic bonds aligned in specific directions within the crystal.
Understanding Unit Cells and Lattice Parameters
The FCC structure can be described using the concept of a unit cell. A unit cell is the smallest repeating unit that, when repeated in three dimensions, generates the entire crystal lattice. In NaCl, the unit cell contains one sodium ion and one chloride ion, although each ion is shared between several adjacent unit cells. The dimensions of this unit cell, including its edge length (lattice parameter), are determined by the ionic radii of Na⁺ and Cl⁻ and the strength of the electrostatic interactions between them.
X-ray Diffraction and Crystal Structure Determination
The precise arrangement of ions within the NaCl crystal lattice was first determined using X-ray diffraction. This technique utilizes the diffraction of X-rays by the crystal lattice to determine the spacing between atoms and ions within the crystal. The resulting diffraction pattern provides a unique fingerprint of the crystal structure, confirming the FCC arrangement of Na⁺ and Cl⁻ ions in sodium chloride. X-ray diffraction remains a vital tool in materials science and crystallography for characterizing the atomic-scale structure of various materials.
Sodium Chloride's Diverse Applications: Beyond the Kitchen Table
While readily recognizable as table salt, sodium chloride's uses extend far beyond seasoning food. Its properties make it an indispensable material in a wide range of industries and applications.
Food Preservation and Flavor Enhancement
The most common use of sodium chloride is as a food preservative. Its ability to draw water out of microorganisms through osmosis inhibits their growth and prevents spoilage. This ancient technique remains crucial for food safety and extending shelf life. Beyond preservation, NaCl provides essential flavor enhancement, adding a distinct salty taste to countless dishes worldwide.
Industrial Applications: From De-icing to Chemical Production
Sodium chloride plays a critical role in various industrial processes. It's used extensively in the de-icing of roads and pavements during winter, lowering the freezing point of water and preventing the formation of ice. In the chemical industry, it's a crucial feedstock for the production of other chemicals, including chlorine gas (Cl₂) and sodium hydroxide (NaOH), both of which have extensive industrial applications.
Medical and Pharmaceutical Uses
Sodium chloride is essential in maintaining the proper electrolyte balance in the body, and solutions of sodium chloride in water (saline solution) are used intravenously to replenish fluids and electrolytes in patients. It's also used in various pharmaceutical preparations as an excipient, a substance that helps in the formulation and administration of medications.
Water Softening and Other Applications
Sodium chloride is a key component in water softening systems. It's used to regenerate ion-exchange resins, removing calcium and magnesium ions from hard water. Beyond these uses, sodium chloride finds applications in leather processing, textiles, and metallurgy, demonstrating its versatility and widespread importance in various industries.
Biological Significance of Sodium Chloride: A Key Electrolyte
Sodium chloride is not merely an industrial chemical; it's a vital component of biological systems. Sodium (Na⁺) and chloride (Cl⁻) ions are crucial electrolytes that play essential roles in numerous physiological processes.
Maintaining Fluid Balance and Osmotic Pressure
Sodium ions contribute significantly to the maintenance of fluid balance and osmotic pressure in cells and tissues. They influence the movement of water across cell membranes, ensuring proper hydration and cell function. An imbalance in sodium levels can lead to various health issues, including dehydration and edema.
Nerve Impulse Transmission and Muscle Contraction
Sodium ions are crucial for nerve impulse transmission and muscle contraction. The movement of sodium ions across nerve cell membranes initiates the electrical signals that transmit information throughout the nervous system. Similarly, sodium ions play a vital role in muscle contraction, enabling the coordinated movements of the body.
Chloride Ions and Gastric Acid Production
Chloride ions (Cl⁻) are also biologically significant. They are essential in the production of gastric acid (HCl) in the stomach, which aids in digestion. Chloride ions also contribute to maintaining proper acid-base balance in the body. Disruptions in chloride ion levels can lead to various health problems.
Conclusion: The Enduring Importance of Sodium Chloride
Sodium chloride, seemingly simple in its chemical formula, reveals a rich tapestry of chemical, physical, and biological significance. From its exemplary demonstration of ionic bonding and its perfectly ordered crystal structure to its diverse applications across various industries and its crucial role in biological systems, NaCl stands as a prime example of a compound whose importance extends far beyond its everyday use as table salt. Understanding the fundamental properties and multifaceted applications of sodium chloride provides valuable insights into the principles of chemistry, materials science, and biology. Its pervasive presence and significance highlight the profound impact of even seemingly simple chemical compounds on our lives and the world around us.
Latest Posts
Latest Posts
-
Which Of The Following Is A Single Replacement Reaction
Apr 03, 2025
-
Point Estimate Of The Population Standard Deviation
Apr 03, 2025
-
A Magnifier Makes Things Appear Larger Because
Apr 03, 2025
-
What Is The Difference Between Intermolecular And Intramolecular Forces
Apr 03, 2025
-
Where Is The Energy Stored In Glucose
Apr 03, 2025
Related Post
Thank you for visiting our website which covers about Sodium Chloride Is An Example Of . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
